
Concept explainers
a.
Interpretation: All the constitutional isomers that have molecular formulas
Concept Introduction: The molecules that possess the same molecular formula but differ in the structural arrangement of atoms in the molecule are said to be isomers of each other.
a.
Answer to Problem 32PP
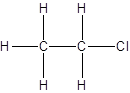
Explanation of Solution
For the first isomer structure, all two-carbon atoms are written in a straight chain and the chlorine atom is bonded to one of the carbon atoms as:
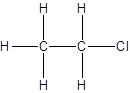
There is no other possibility for the different structural formula
b.
Interpretation: All the constitutional isomers that have molecular formulas
Concept Introduction: The molecules that possess the same molecular formula but differ in the structural arrangement of atoms in the molecule are said to be isomers of each other.
b.
Answer to Problem 32PP
Isomer I:
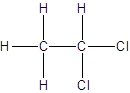
Isomer II:
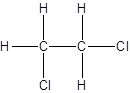
Explanation of Solution
The different structural the arrangement of atoms in the molecule
- All two-carbon atoms are written in a straight chain and two chlorine atoms are bonded to one of the carbon atoms resulting in:
- Interchanging the position of one hydrogen atom at first carbon with one chlorine atom of the second carbon atom.
Isomer I:
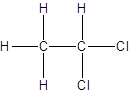
Isomer II:
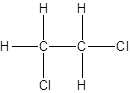
c.
Interpretation: All the constitutional isomers that have molecular formulas
Concept Introduction: The molecules that possess the same molecular formula but differ in the structural arrangements of atoms in the molecule are said to be isomers of each other.
c.
Answer to Problem 32PP
Isomer I:
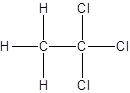
Isomer II:
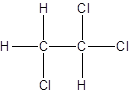
Explanation of Solution
The different structural arrangement of atoms in the molecule
- All two-carbon atoms are written in a straight chain and three chlorine atoms are bonded to one of the carbon atoms resulting in:
- Interchanging the position of one hydrogen atom at first carbon with one chlorine atom of the second carbon atom.
Isomer I:
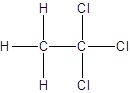
Isomer II:
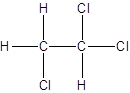
d.
Interpretation: All the constitutional isomers that have molecular formulas
Concept Introduction: The molecules that possess the same molecular formula but differ in the structural arrangements of atoms in the molecule are said to be isomers of each other.
d.
Answer to Problem 32PP
Isomer I:
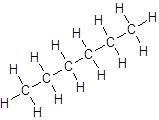
Isomer II:
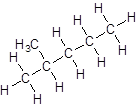
Isomer III:
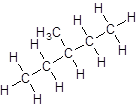
Isomer IV:
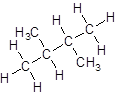
Isomer V:
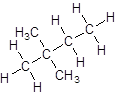
Explanation of Solution
The different structural arrangement of atoms in the molecule
- All six-carbon atoms are written in a straight chain resulting in:
- Five-carbon atoms are written in a straight chain and a methyl group is attached to the second carbon resulting in:
- Five-carbon atoms are written in a straight chain and a methyl group is attached to the third carbon resulting in:
- Four-carbon atoms are written in a straight chain and a methyl group is attached to the second and third carbon resulting in:
- Four-carbon atoms are written in a straight chain and two methyl groups are attached to the second carbon resulting in:
Isomer I:
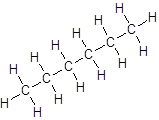
Isomer II:
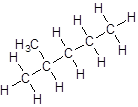
Isomer III:
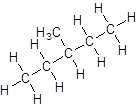
Isomer IV:
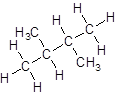
Isomer V:
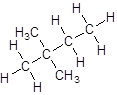
Want to see more full solutions like this?
Chapter 1 Solutions
ORGANIC CHEMISTRY,SOLNS...-ETEXT+BOX
- 7.35 Which one of the following contains botb ionic and covalent bonds? (a) BaCO3 , (b) MgCl2 , (c) BaO , (d) H2S , (e) So42arrow_forwardA. Propane Measurements: C1–C2–C3 bond angle 1. H–C2–H bond angle 2. H–C1–C2 bond angle 3. H–C1–H bond angle 4. C–C bond length in propane 5. B. 2,2-Dimethylbutane Measurements: Carbon numbering here refers to IUPAC numberings. C2–C3–C4 bond angle 6. anti-C1–C2–C3 bond angle 7. gauche-C1–C2–C3 bond angle 8. C2–C3 bond length 9. C. Butane Measurements: Strain energy—anti conformation 10. C1–C2–C3 bond angle—anti 11. H–C1–C2 bond angle—anti 12. DAtA & AnALYSiS Molecular Modeling I 75 C1–C2–C3–H dihedral angle—anti 13. Strain energy—gauche conformation 14. C1–C2–C3 bond angle—gauche 15. C1–C2–C3–C4 dihedral angle—gauche 16. C1–C4 H–H distance—gauche 17. H–C1–C2–C3 dihedral angle—gauche 18. PART II. BOND…arrow_forward26.) How many sigma and how many pi bonds are there in molecule below? H3C-CH=CH-CH3 Write your answer as _____ sigma and _____ pi. For example, 6 sigma and 7 pi (no period at end).arrow_forward
- Answer the following question with reference to the molecule below: 61 H3C H (a) Label the shortest C–C single bond. (b) Label the longest C–C single bond. (c) Considering all the bonds, label the shortest C–C bond (d) Label the strongest C–H bond. (e) Account for the difference in bond lengths for C4–C5 and C5–C6, even though they are both C-C single bonds.arrow_forwardCalculate the dihedral angle of a molecule where the bonds are as follows: C1-C2: 1.60 A C2-C3: 1.25 A C3-C4: 1.60 A All C-C-C bond angles are 125 degrees. Explain.arrow_forwardWhat is the chemical formula for the molecule represented by the skeletal structure below? = Select one: a. Cannot be determined from this skeletal structure b. C2 c. C2H2 d. CH4arrow_forward
- Rank the bonds below from most polar to least polar A I>II>III>IV B IV>III>II>I C II>I>IV>III D I>III>II>IV E I>IV>III>IIarrow_forward49. Among the three structures below for CO2 : (1) O-C=O (2) O=C-O (3) O-C-O (4) O=C=O Which is the best structure? Select one: a. (1) and (2) b. (2) and (4) c. (4) d. (3)arrow_forwardShow the expanded structure (draw all the bonds and lone pairs) of Levothyroxine. Determine the molecular geometry of each central atom. Color each central atom based on its molecular geometry and include a key for your colors. For example, color tetrahedral C’s red, trigonal planar C’s , blue, etc.arrow_forward
- I need helping solving these and their are 2 more questions within this problem as well but I couldn't upload all The last two are: What is the C−C−C bond angle in propene? 109.5° 180° 90° 120° Does propene have an overall two‑dimensional shape or three‑dimensional shape? three‑dimensional two‑dimensionalarrow_forwardFind the empircal formula of a compound made of 93.74%C and 6.26%H.arrow_forward5. Use you knowledge of molecular structure and bonding to explain why pentane (C5H12) higher boiling point than 2,2- dimethylpropane (C5H12).arrow_forward
 Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning




