
ORGANIC CHEM PRINT STUDY GDE & SSM
4th Edition
ISBN: 9781119810650
Author: Klein
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Question
Chapter 1, Problem 48PP
Interpretation Introduction
Interpretation: The total number of
bonds should be counted in the following compound:
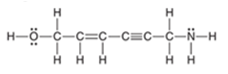
Concept Introduction:
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
t
Describe each highlighted bond in terms of the overlap of atomic orbitals.
(If the highlighted bond is not a pi bond, select the blank option from the dropdown menu.)
Compound 1:
Η Η Η
H-C-C-CN
HHH H
Molecular orbital type:
Atomic orbitals in the sigma bond:
Atomic orbitals in the
Compound 2:
bond:
HH
H-C-C-C
HH
H
EN-H
H
Molecular orbital type:
Atomic orbitals in the sigma bond:
Atomic orbitals in the pi bond:
3) How many electrons are delocalized in how many p-orbitals?
Draw the most and second most stable resonance structures of the following compound.
Order the structures by increasing stability. What is the partial charge distribution in the
resonance hybrid?
Draw the skeletal (bond-line) structures of the possible isomers of C₃H₅Cl including configurational isomers.
BOND-LINE STRUCTURE ONLY PLEASE
Chapter 1 Solutions
ORGANIC CHEM PRINT STUDY GDE & SSM
Ch. 1.2 - Prob. 1LTSCh. 1.2 - Prob. 2ATSCh. 1.2 - Prob. 2LTSCh. 1.3 - Prob. 3LTSCh. 1.3 - Prob. 4PTSCh. 1.3 - Prob. 5PTSCh. 1.4 - Prob. 4LTSCh. 1.4 - Prob. 7PTSCh. 1.4 - Prob. 8PTSCh. 1.4 - Prob. 9ATS
Ch. 1.5 - Prob. 5LTSCh. 1.5 - Prob. 10PTSCh. 1.5 - Prob. 11ATSCh. 1.5 - Prob. 12ATSCh. 1.6 - Prob. 6LTSCh. 1.6 - Prob. 14ATSCh. 1.7 - Prob. 7LTSCh. 1.7 - Prob. 17ATSCh. 1.10 - Prob. 18CCCh. 1.10 - Prob. 20CCCh. 1.10 - Prob. 8LTSCh. 1.10 - Prob. 21PTSCh. 1.10 - Nemotin is a compound that was first isolated from...Ch. 1.10 - Prob. 23CCCh. 1.11 - Prob. 9LTSCh. 1.11 - Prob. 24PTSCh. 1.11 - Prob. 25PTSCh. 1.11 - Prob. 26PTSCh. 1.11 - Prob. 27ATSCh. 1.12 - Prob. 10LTSCh. 1.12 - Prob. 29ATSCh. 1.13 - Prob. 11LTSCh. 1.13 - Prob. 31ATSCh. 1 - Prob. 32PPCh. 1 - Prob. 33PPCh. 1 - Prob. 34PPCh. 1 - Prob. 35PPCh. 1 - Prob. 36PPCh. 1 - Prob. 37PPCh. 1 - Prob. 38PPCh. 1 - Prob. 39PPCh. 1 - Prob. 40PPCh. 1 - Prob. 41PPCh. 1 - Prob. 42PPCh. 1 - Prob. 44PPCh. 1 - Prob. 45PPCh. 1 - Prob. 46PPCh. 1 - Prob. 47PPCh. 1 - Prob. 48PPCh. 1 - Prob. 49PPCh. 1 - Prob. 50PPCh. 1 - Prob. 51PPCh. 1 - Prob. 52PPCh. 1 - Prob. 53PPCh. 1 - Prob. 54PPCh. 1 - Nicotine is an addictive substance found in...Ch. 1 - Prob. 56PPCh. 1 - Prob. 57PPCh. 1 - Prob. 59PPCh. 1 - Prob. 63ASPCh. 1 - Prob. 64ASPCh. 1 - Prob. 66ASPCh. 1 - Prob. 69ASPCh. 1 - Prob. 71ASPCh. 1 - Prob. 72ASPCh. 1 - Prob. 75IP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Identify the hybridization state, molecular geometry and approximate bond angle at the indicatednitrogen atom in the following compound. O sp²,bent, 109° O sp²,bent, 120° O sp3, tetrahedral, 109.5° O sp3, trigonal pyramidal, ~109.5° O sp2, trigonal pyramidal, 120° CH3 HH CH₂. H₂=C₂C=C₂C₂O₂H N OU! H³N¬C-C¬-H H HH .0.arrow_forwardIn the sketch of the structure of SO2 label all bonds. Drag the appropriate labels to their respective targets. Labels can be used once, more than once, or not at all. : S(p) – O(p) Lone pair in p orbital Lone pair in sp? orbital o : S(p) – 0(sp²) т: S(p) — О(p) T: S(sp²) – O(p) r: S(sp²) – O(p) S(p) – O(sp²)arrow_forward6. On the basis of molecular orbital theory and Hückel's rule, which molecules and/or ions should be aromatic? I VI II VII X HH E III Η Η VIII IV + H IX Varrow_forward
- Draw the bond-line (skeletal) formula of a compound with the molecular formula C5H10Cl2 that meets the specified condition.arrow_forwardß-carotene is an antioxidant and a precursor to vitamin A found in carrots and other brightly colored foods. How many π bonds are present in the structure of ß-carotene shown? CH₂ CH₂ H₂C H₂C D CH₂arrow_forwardEvaluate the type of molecular bonding (sigma or pi) in a covalent molecule. (Z,Z)-3,6-nonadienal is the molecule responsible for the aroma of watermelon. How many sigma and pi bonds are in this compound, C9H140? A structural formula is here: CH3-CH2-CH=CH-CH2-CH=CH-CH2- CH=O and a structure is shown below H H H H H H Harrow_forward
- The unsaturation number or degree of unsaturation (U) can be used to determine the number of rings and multiple bonds in a compound from its molecular formula. Given a structure, you can determine the number of hydrogens without having to count them explicitly. Consider three compounds and their degree of unsaturation. (a) A compound A has the molecular formula C7H13ClN2OC7H13ClN2O. How many rings and/or π bonds does it contain?arrow_forwardCalculate the charge distribution and bond orders of 2,3-dimethyl butadiene.arrow_forwardDraw a bond-line structure for the following:arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
Linear Combination of Atomic Orbitals LCAO; Author: Edmerls;https://www.youtube.com/watch?v=nq1zwrAIr4c;License: Standard YouTube License, CC-BY
Quantum Molecular Orbital Theory (PChem Lecture: LCAO and gerade ungerade orbitals); Author: Prof Melko;https://www.youtube.com/watch?v=l59CGEstSGU;License: Standard YouTube License, CC-BY