
ORGANIC CHEMISTRY - CONNECT ACCESS CARD
10th Edition
ISBN: 9781259636394
Author: Carey
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 1, Problem 57P
The structure of montelukast, an antiasthma drug, is shown here.
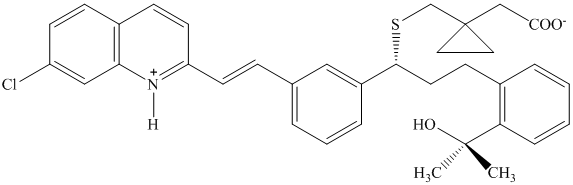
Use Table
to identify the most acidic and most basic sites in the molecule.
(Although you won’t find an exact match in structure, make a prediction based on
analogy with similar groups in simpler molecules.)
Write the structure of the product formed by treating montelukast with one equivalent
of sodium hydroxide.
Write the structure of the product formed by treating montelukast with one equivalent
of HCl.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
What is the conjugate acid of each of the following? What is the conjugate base of each? (Omit states-of-matter from your answer.)
H2S
Conjugate acid=
Conjugate base =
H2PO4−
Conjugate acid=
Conjugate base =
HSO3−
Conjugate acid=
Conjugate base =
Organic Chemistry 1
Why did you choose your choice? Explain your reasoning and rational (theory and principles and periodic trends) as to why the species is the strongest base or the strongest conjugate acid. That is to say, provide a reasoning and an answer to your selection/choice. Why did you circle your answer for that particular species?
Can someone help me find the most acidic compound out of these two compounds? Thank you!
Chapter 1 Solutions
ORGANIC CHEMISTRY - CONNECT ACCESS CARD
Ch. 1.1 - How many electrons does carbon have? How many are...Ch. 1.1 - Referring to the periodic table as needed, write...Ch. 1.2 - Species that have the same number of electrons are...Ch. 1.2 - Which of the following ions possess a noble gas...Ch. 1.2 - Prob. 5PCh. 1.3 - Prob. 6PCh. 1.3 - Problem 1.7 All of the hydrogens are bonded to...Ch. 1.4 - Problem 1.8 In which of the compounds...Ch. 1.4 - Indicate the direction of the dipole for the...Ch. 1.5 - Prob. 10P
Ch. 1.5 - The following inorganic species will be...Ch. 1.5 - Prob. 12PCh. 1.6 - Prob. 13PCh. 1.6 - Problem 1.14 Nitrosomethane and formaldoxime both...Ch. 1.6 - Prob. 15PCh. 1.7 - All of the bonds in the carbonate ion (CO32-) are...Ch. 1.7 - Prob. 17PCh. 1.8 - Prob. 18PCh. 1.8 - Prob. 19PCh. 1.9 - Sodium borohydride, NaBH4, has an ionic bond...Ch. 1.9 - Prob. 21PCh. 1.10 - Which of the following compounds would you expect...Ch. 1.11 - Using the curved arrow to guide your reasoning,...Ch. 1.11 - Prob. 24PCh. 1.11 - Prob. 25PCh. 1.12 - Prob. 26PCh. 1.12 - Prob. 27PCh. 1.12 - Prob. 28PCh. 1.12 - Prob. 29PCh. 1.12 - Prob. 30PCh. 1.13 - Which is the stronger acid, H2O or H2S? Which is...Ch. 1.13 - Prob. 32PCh. 1.13 - Prob. 33PCh. 1.13 - Hypochlorous and hypobromous acid (HOClandHOBr)...Ch. 1.13 - Prob. 35PCh. 1.13 - Prob. 36PCh. 1.14 - What is the equilibrium constant for the following...Ch. 1.14 - Prob. 38PCh. 1.14 - Prob. 39PCh. 1.15 - Write an equation for the Lewis acid/Lewis base...Ch. 1 - Write a Lewis formula for each of the following...Ch. 1 - Prob. 42PCh. 1 - Write structural formulas for all the...Ch. 1 - Prob. 44PCh. 1 - Expand the following structural representations so...Ch. 1 - Each of the following species will be encountered...Ch. 1 - Consider Lewis formulas A, B, and C: H2 C -NN:...Ch. 1 - Prob. 48PCh. 1 - Prob. 49PCh. 1 - Prob. 50PCh. 1 - Prob. 51PCh. 1 - Prob. 52PCh. 1 - Prob. 53PCh. 1 - Prob. 54PCh. 1 - Which compound in each of the following pairs...Ch. 1 - With a pKa of 11.6, hydrogen peroxide is a...Ch. 1 - The structure of montelukast, an antiasthma drug,...Ch. 1 - One acid has a pKa of 2, the other has a pKa of 8....Ch. 1 - Calculate Ka for each of the following acids,...Ch. 1 - Rank the following in order of decreasing acidity....Ch. 1 - Rank the following in order of decreasing...Ch. 1 - Consider 1.0 M aqueous solutions of each of the...Ch. 1 - Prob. 63PCh. 1 - Prob. 64PCh. 1 - Prob. 65PCh. 1 - Prob. 66PCh. 1 - Prob. 67PCh. 1 - Prob. 68PCh. 1 - Amide Lewis Structural Formulas Lewis formulas are...Ch. 1 - Amide Lewis Structural Formulas Lewis formulas are...Ch. 1 - Amide Lewis Structural Formulas Lewis formulas are...Ch. 1 - Prob. 72DSPCh. 1 - Amide Lewis Structural Formulas Lewis formulas are...Ch. 1 - Amide Lewis Structural Formulas Lewis formulas are...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- The following are equivalent ways of asking about the acidity of an H atom: • What is the most acidic H on the molecule? • Which H is associated with the published pKa value? • Which H on the molecule is easiest to remove? • Which H on the molecule takes the least energy to remove? • Which bond to an H is most polarized? • For which H atom is removal least uphill in energy? • Which bond to an H atom, when broken, results in the lowest PE conjugate base? We will often find the last of these questions is easiest to answer. To do this, find all the different Hatoms on the molecule, and draw all possible conjugate bases.Only the lowest-energy one is the “real” conjugate base. Identify this structure, and you have found the most acidic H. Use this strategy to find the most acidic H on each of the following molecules. Note: Each structure hasat least three different kinds of H’s, so draw at least three unique conjugate bases for each.arrow_forwardCan you show me how to solve this question I found in my review?arrow_forwardUsing the data in the table, which of the conjugate bases below is the strongest base? A) OAc- B) C7H5O2- C) NO2- D) F- E) OAc- and C7H5O2-arrow_forward
- Please use the values in the resources listed below instead of the textbook values. What is the conjugate acid of each of the following? What is the conjugate base of each? (Omit states-of-matter from your answer.) (a) HNO2 Conjugate acid Conjugate base (b) C6H5OH Conjugate acid Conjugate base (c) H5N2+ Conjugate acid Conjugate basearrow_forwardDraw the most stable conjugate base of this molecule. Briefly explain your choice in thespace provided.arrow_forwardWhat is the conjugate base of H2C6H5O7-? H3C6H6O7 H3C6H5O73- None of these HC6H5O72- C6H5O73-arrow_forward
- a. Which of the acids in the table above has the strongest conjugate base? b. What is the structure of the strongest acid in the table above?arrow_forwardThe answer is D (not C). The explanation given is "This compound is the only one of the choices that does not contain a base." What does it mean by "does not contain a base"? Isn't Br- a base?arrow_forwardIn the reaction NH2- + CH3OH → NH3 + CH3O-, Bronsted would consider NH2- to be the (acid or base?) CH3OH to be the (acid or base?) , NH3 to be the (conjugate acid or conjugate base?) , and CH3O- to be the (conjugate acid or conjugate base?)arrow_forward
- Draw the conjugate base and two resonance structures of the most acidic compoundarrow_forwardSelect the stronger acid from pair and explain your reasoning. For stronger acid, write a structural formula for its conjugate base. Q.) H2O or H2CO3arrow_forwardConsider cyclohexane and benzene (draw the structures below). Draw the conjugate base of each molecule and then explain why cyclohexane is the weaker acid.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
General Chemistry | Acids & Bases; Author: Ninja Nerd;https://www.youtube.com/watch?v=AOr_5tbgfQ0;License: Standard YouTube License, CC-BY