
Interpretation: The reason corresponding to the fact that the permanent dipole moment of 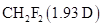 is larger than the permanent dipole moment of
is larger than the permanent dipole moment of 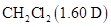 , yet the boiling point of
, yet the boiling point of 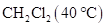 is much higher than that of
is much higher than that of 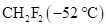 is to be stated.
is to be stated.
Concept introduction: The intermolecular interaction between non-polar or least polar molecules is known as dispersion force. This is also known as, London force in honor of the Fritz London’s great work on it. The strength of this force depends upon the surface area of the molecule. The strength of force is directly proportional to the surface area.
To determine: The reason corresponding to the fact that the permanent dipole moment of 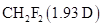 is larger than the permanent dipole moment of
is larger than the permanent dipole moment of 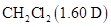 , yet the boiling point of
, yet the boiling point of 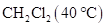 is much higher than that of
is much higher than that of 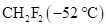 .
.
Want to see the full answer?
Check out a sample textbook solution
Chapter 10 Solutions
Chemistry: The Science in Context (Fifth Edition)
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





