![OWLv2 for Bettelheim/Brown/Campbell/Farrell/Torres' Introduction to General, Organic and Biochemistry, 11th Edition, [Instant Access], 1 term (6 months)](https://s3.amazonaws.com/compass-isbn-assets/textbook_empty_images/large_textbook_empty.svg)
OWLv2 for Bettelheim/Brown/Campbell/Farrell/Torres' Introduction to General, Organic and Biochemistry, 11th Edition, [Instant Access], 1 term (6 months)
11th Edition
ISBN: 9781305106734
Author: Frederick A. Bettelheim; William H. Brown; Mary K. Campbell; Shawn O. Farrell; Omar Torres
Publisher: Cengage Learning US
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 10, Problem 10.24P
10-24 Use the VSEPR model to predict the bond angles and geometry about each highlighted atom. (Hint-. Remember to take into account the presence of unshared pairs of electrons.) H H
I I
- H-C-C-O-H I I H H
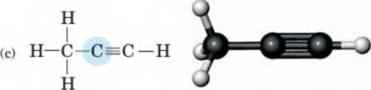
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
4. Convert the following structures into bond line structures.
C-H
ннн
нон
нн
Н-с-с-с-о-н
Н-с-с-с—Н
Н-с-с-о-с-н
H H H
H H H
нн
H C.
H-
H-N H Ó H
il
н-с-с-с-с-н
H H H H
нн
XOVW3_VV
u-IgNslkr7j8P3jH-liJQOWQJvqYPWI
Determine the relationship between Structure A and Structure B in each row of the table.
Structure A
Structure B
Relationship
o isomers
H.
:0:
H
H
D-H
H-C=C-C-C-H
H-C= C-C= C-H
O resonance structures
H.
H
H
O neither
o isomers
:S-c=N:
:NEC-S:
O resonance structures
o neither
o isomers
H-N-C=N:
O resonance structures
H.
H-C=
O neither
75°F
DELL
Using Newman projections, choose the most stable
conformer for each of the following:
3-methylpentane, viewed along the C-2 - C-3 bond
CH3
H
CH2CH3
H
H
CH3
H
H.
CH2CH3
H
CH3
CH3
CH
H
CH
H
H
CH₂CH,
Chapter 10 Solutions
OWLv2 for Bettelheim/Brown/Campbell/Farrell/Torres' Introduction to General, Organic and Biochemistry, 11th Edition, [Instant Access], 1 term (6 months)
Ch. 10.3 - Prob. 10.1PCh. 10.4 - Prob. 10.2PCh. 10.4 - Prob. 10.3PCh. 10.4 - Prob. 10.4PCh. 10.4 - Prob. 10.5PCh. 10.4 - Prob. 10.6PCh. 10 - Prob. 10.7PCh. 10 - Prob. 10.8PCh. 10 - 10-9 Is there any difference between vanillin made...Ch. 10 - Prob. 10.10P
Ch. 10 - 10-11 What important experiment did Wohler carry...Ch. 10 - Prob. 10.12PCh. 10 - Prob. 10.13PCh. 10 - Prob. 10.14PCh. 10 - 10-15 How many electrons are in the valence shell...Ch. 10 - 10-16 What is the relationship between the number...Ch. 10 - Prob. 10.17PCh. 10 - Prob. 10.18PCh. 10 - 10-19 Write Lewis structures for these ions. (a)...Ch. 10 - 10-20 Why are the following molecular formulas...Ch. 10 - 10-21 Explain how to use the valence-shell...Ch. 10 - 10-22 Suppose you forget to take into account the...Ch. 10 - Suppose you forget to take into account the...Ch. 10 - 10-24 Use the VSEPR model to predict the bond...Ch. 10 - Prob. 10.25PCh. 10 - Prob. 10.26PCh. 10 - 10-27 What is meant by the term functional group?Ch. 10 - 10-28 List three reasons why functional groups are...Ch. 10 - Prob. 10.29PCh. 10 - Prob. 10.30PCh. 10 - Prob. 10.31PCh. 10 - 10-32 Draw a structural formula for the one...Ch. 10 - 10-33 What is the meaning of the term tertiary (...Ch. 10 - Prob. 10.34PCh. 10 - Draw structural formulas for each of the...Ch. 10 - 10-36 Draw structural formulas for the six ketones...Ch. 10 - 10-37 Draw structural formulas for the eight...Ch. 10 - Prob. 10.38PCh. 10 - 10-39 (Chemical Connections 10A) How was Taxol...Ch. 10 - Prob. 10.40PCh. 10 - Prob. 10.41PCh. 10 - Silicon is immediately below carbon in Group 4A of...Ch. 10 - 10-43 Phosphorus is immediately below nitrogen in...Ch. 10 - Draw the structure for a compound with the...Ch. 10 - 10-45 Draw structural formulas for the eight...Ch. 10 - Prob. 10.46PCh. 10 - 10-47 Which of these covalent bonds are polar, and...Ch. 10 - Of the bonds in Problem 10-47, which is the most...Ch. 10 - Prob. 10.49PCh. 10 - Prob. 10.50PCh. 10 - Following is a structural formula for naphthalene....Ch. 10 - Prob. 10.52PCh. 10 - Prob. 10.53PCh. 10 - Urea, (NH.,)2CO, is used in plastics and in fertil...Ch. 10 - Prob. 10.55PCh. 10 - Prob. 10.56PCh. 10 - Aspirin is prepared by the reaction of salicylic-...Ch. 10 - Following is the structural formula of acetamide....Ch. 10 - Prob. 10.59P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 10-21 Explain how to use the valence-shell electron-pair repulsion (VSEPR) model to predict bond angles and geometry about atoms of carbon, oxygen, and nitrogen.arrow_forward10-43 Phosphorus is immediately below nitrogen in Group 5A of the Periodic Table. Predict the C—P—C bond angles in trimethylphosphine, (CHaJaP.arrow_forward10-22 Suppose you forget to take into account the presence of the unshared pair of electrons on nitrogen in the molecule NH3. What would you then predict for the H—N—H bond angles and the geometry (bond angles and shape) of ammonia?arrow_forward
- 1:54 AM Wed May 18 8 Q 口山 EC Organic Chemistry 203 - 8. a) State the hybridization (sp°, sp², sp) of each carbon in this molecule, going left to right. N=CCH2CH2OH b) Write "most" under the most stable alkene. Write "least" under the least stable alkene. CH3 CH3 CH3 CH2 H;C HC H3C c) Write "most" under the most stable cation. Write "least" under the least stable cation. H3C CH3 CH3 CH3 CH3 H3C H3C H3Carrow_forwardThe following bonds in vinyl alcohol, what is the bond angles for C-O-H? See the structure as your basis. ... нн H-c=c-ö-H A) 109.5° B) 90° c) 180° D) 120°arrow_forwardHow many o and bonds are in this molecule? TO Ö-H H H-C C-C || H C=C number of o bonds: C-H H MacBook Pro number of bonds:arrow_forward
- b Which atom in the following structure has the incorrect number of bonds? нн Cl-c=c-H H. нн HH Cifc+c-H HH Ci-c=c-H H. нн Ci-c=c-Harrow_forwardCl ledu.co Maps Q A Bb BI CH :0 O F1 CH ►Ch Ac Be G dc B ky o Ma b Hc G 17 Question 7 of 48 Provide the correct IUPAC name for the skeletal (line-bond) structure shown here. 3,3,3,3- 2,2,3,3- 2,3-3,3- 2,2,2,2- 2,2- tri penta tetra di 2 0³ F2 W S 3 E 80 F3 D $ 4 ♂ F4 R F do L % 5 2,2,3,3-2,3- 16 뽀 F5 T X G prop hex eth pent but meth ene vne ane MacBook Air C F6 6 Y H & 7 F7 [S U * 8 J W DII F8 1 ( 9 K F9 0 0 L C S UO NE 7 F10 P F11 { [arrow_forwardFor each example, specify whether the two structures are resonance contributors to the same resonance hybrid. CH2 HC a) HC=CH2 HC-CH2 HC-CH2 ö: ö: b) H3C- H3C- NH2 NH2 c) H3C-NH-CH2 H3C-NH=CH2arrow_forward
- Chemistry For each example, specify whether the two structures are resonance contributors to the same resonance hybrid. ő: :0: a) H₂C- -NH₂ H₂C- HỘ CHỊCHÍCH, ő: H₂C=CH-CH b) H 20-CH₂ CH₂ CH₂ :0: H₂C-CH=C-Harrow_forwardQ1: Use the &+ or 8- convention to indicate the direction of expected polarity for every polar bond in the following molecules. Predict the dipole moment and show its direction by using crossed arrow (+). но. N. OH CH3arrow_forwardНЬ C-Hd O C-Hb C-Hc Which C-H bond has the lowest BDE? C-Ha O C-He Ha He Hc -Hdarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning

Acid-Base Titration | Acids, Bases & Alkalis | Chemistry | FuseSchool; Author: FuseSchool - Global Education;https://www.youtube.com/watch?v=yFqx6_Y6c2M;License: Standard YouTube License, CC-BY