
ORGANIC CHEMISTRY STD. GDE W/CONNECT PK
5th Edition
ISBN: 9781260107333
Author: SMITH
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 10, Problem 10.33P
(a) Label the carbon-carbon double bond in A as
stereoisomers) formed when A is treated with
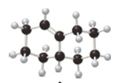
A
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Draw the organic products formed when cyclopentene is treated withfollowing reagent.
Na, NH3
Draw the structure of cholic acid, showing the rings in their chairconformations, and label each methyl group and hydroxy group asaxial or equatorial.
4 Answer the following questions about compound B, represented in the given ball-and-stick model. a. Convert B to a condensed structure and give its IUPAC name. b. What product is formed when B is treated with H 2 in the presence of a metal catalyst? c. What product is formed when B is treated with H 2O in the presence of H 2SO 4? d. What polymer is formed when B is polymerized?
Chapter 10 Solutions
ORGANIC CHEMISTRY STD. GDE W/CONNECT PK
Ch. 10 - Prob. 10.1PCh. 10 - Problem 10.2 How many degrees of unsaturation are...Ch. 10 -
Problem 10.3 How many degrees of unsaturation...Ch. 10 - Give the IUPAC name for each alkene. abcdeCh. 10 - Give the IUPAC name for each polyfunctional...Ch. 10 - Problem 10.6 Label each C-C double bond as E or Z....Ch. 10 - Prob. 10.7PCh. 10 - Prob. 10.8PCh. 10 - Prob. 10.9PCh. 10 - Problem 10.10 Rank the following isomers in order...
Ch. 10 - Linolenic acidTable 10.2 and stearidonic acid are...Ch. 10 - Prob. 10.12PCh. 10 - Problem 10.13 What product is formed when each...Ch. 10 - Prob. 10.14PCh. 10 - Problem 10.15 Draw the products formed when each...Ch. 10 - Prob. 10.16PCh. 10 - Prob. 10.17PCh. 10 - Addition of HBr to which of the following alkenes...Ch. 10 - Problem 10.19 Draw the products, including...Ch. 10 - Prob. 10.20PCh. 10 - Problem 10.21 What two alkenes give rise to each...Ch. 10 - Prob. 10.22PCh. 10 - Problem 10.23 Draw the products of each reaction,...Ch. 10 - Problem 10.24 Draw all stereoisomers formed in...Ch. 10 - Prob. 10.25PCh. 10 - Problem 10.26 What alkylborane is formed from...Ch. 10 - Draw the products formed when each alkene is...Ch. 10 - What alkene can be used to prepare each alcohol as...Ch. 10 - Prob. 10.29PCh. 10 - Draw the products of each reaction using the two...Ch. 10 - Problem 10.31 Devise a synthesis of each compound...Ch. 10 - Give the IUPAC name for each compound. a.b.Ch. 10 - a Label the carbon-carbon double bond in A as E or...Ch. 10 - Prob. 10.34PCh. 10 - 10.35 Calculate the number of degrees of...Ch. 10 - Prob. 10.36PCh. 10 - Label the alkene in each drug as E or Z....Ch. 10 - Give the IUPAC name for each compound. a. c. e. b....Ch. 10 - Prob. 10.39PCh. 10 - 10.40 (a) Draw all possible stereoisomers of, and...Ch. 10 - Prob. 10.41PCh. 10 - 10.42 Now that you have learned how to name...Ch. 10 - Prob. 10.43PCh. 10 - Prob. 10.44PCh. 10 - Prob. 10.45PCh. 10 - Draw the products formed when (CH3)2C=CH2 is...Ch. 10 - What alkene can be used to prepare each alkyl...Ch. 10 - Prob. 10.48PCh. 10 - Draw the constitutional isomer formed in each...Ch. 10 - Prob. 10.50PCh. 10 - Draw all stereoisomers formed in each reaction. a....Ch. 10 - Draw the products of each reaction, including...Ch. 10 - Prob. 10.53PCh. 10 - Prob. 10.54PCh. 10 - Prob. 10.55PCh. 10 - 10.56 Draw a stepwise mechanism for the following...Ch. 10 - Prob. 10.57PCh. 10 - Draw a stepwise mechanism for the conversion of...Ch. 10 - Draw a stepwise mechanism that shows how all three...Ch. 10 - Less stable alkenes can be isomerized to more...Ch. 10 - Prob. 10.61PCh. 10 - Prob. 10.62PCh. 10 - Bromoetherification, the addition of the elements...Ch. 10 - Devise a synthesis of each product from the given...Ch. 10 - 10.65 Draw a synthesis of each compound from...Ch. 10 - 10.66 Explain why A is a stable compound but B is...Ch. 10 - Prob. 10.67PCh. 10 - Prob. 10.68PCh. 10 - 10.69 Lactones, cyclic esters such as compound A,...Ch. 10 - 10.70 Draw a stepwise mechanism for the following...Ch. 10 - 10.71 Like other electrophiles, carbocations add...Ch. 10 - 10.72 Draw a stepwise mechanism for the...
Additional Science Textbook Solutions
Find more solutions based on key concepts
Write a Lewis formula for each of the following organic molecules: C2H3Cl (vinyl chloride: starting material fo...
Organic Chemistry - Standalone book
Draw a Lewis structure for each of the following species: a. H2CO3 b. CO32 c. CH2O d. CO2
Essential Organic Chemistry (3rd Edition)
Label each statement about the polynucleotide ATGGCG as true or false. The polynucleotide has six nucleotides. ...
General, Organic, & Biological Chemistry
What is the pH range for acidic solutions? For basic solutions?
EBK INTRODUCTION TO CHEMISTRY
4.1 Write the symbols for the following elements.
a. copper
b. platinum
c. calcium
d. manganese
e. Iron
...
Chemistry: An Introduction to General, Organic, and Biological Chemistry (13th Edition)
Q2. Which statement best defines chemistry?
a. The science that studies solvents, drugs, and insecticides
b. Th...
Introductory Chemistry (6th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Eleostearic acid, C18H30O2, is a rare fatty acid found in the tung oil used for finishing furniture. On ozonolysis followed by treatment with zinc, eleostearic acid furnishes one part pentanal, two parts glyoxal (OHC-CHO), and one part 9-oxononanoic acid [OHC(CH2)7CO2H]. What is the structure of eleostearic acid?arrow_forwardCH3-CH2-OH reacts with H+/140C = ? + H2Oarrow_forwardDraw propyne and fill in all H’s .arrow_forward
- Draw the organic products formed when each compound is treated with H2, Pd-C. Indicate the three-dimensional structure of all stereoisomers formed.arrow_forwardDraw the products formed when each compound is treated with CH3CH2COCl, AlCl3.arrow_forwardCarnauba wax, used in floor and furniture polishes, contains an ester of a C32 straight-chain alcohol with a C20 straight-chain carboxylic acid. Draw its structure.arrow_forward
- A-carrow_forwardStearic acids is used in the production of detergents, soaps and cosmetics because they serves as_________________.I. emulsifierII. emollientIII. lubricantsarrow_forward(a) Draw all possible stereoisomers of 4-methylnon-2-ene, and name each isomer, including its E,Z and R,S prexes. (b) Label two pairs of enantiomers. (c) Label four pairs of diastereomers.arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you

Chapter 4 Alkanes and Cycloalkanes Lesson 2; Author: Linda Hanson;https://www.youtube.com/watch?v=AL_CM_Btef4;License: Standard YouTube License, CC-BY
Chapter 4 Alkanes and Cycloalkanes Lesson 1; Author: Linda Hanson;https://www.youtube.com/watch?v=PPIa6EHJMJw;License: Standard Youtube License