
Give the IUPAC name for each compound.
a.
 e.
e.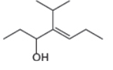
b. 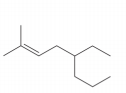 d.
d. 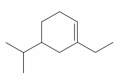 f.
f.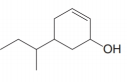
(a)
Interpretation: The IUPAC name for the given compound is to be stated.
Concept introduction: The systematic naming of organic compound is given by IUPAC nomenclature. The naming of organic compound is done such that the structure of organic compound is correctly interpreted from the name.
Rules for writing IUPAC name from structural formula are
1. First identify the longest carbon chain.
2. The next step is to identify the groups attached to the longest chain.
3. Identify the position, location, and number of the substituent bonded to the carbon chain.
4. Use prefix di, tri, tetra if same type of substituent is present.
5. Name the substituents in alphabetical order.
Answer to Problem 10.40P
The IUPAC name for the given compound is
Explanation of Solution
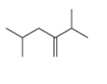
The given compound is
The numbering of carbon atoms present in the longest carbon chain is shown below.
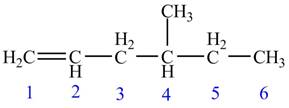
Figure 1
The longest carbon chain has six carbon atoms. The root word used for six carbon atoms is hex and the suffix used for
The IUPAC name for the given compound is
(b)
Interpretation: The IUPAC name for the given compound is to be stated.
Concept introduction: The systematic naming of organic compound is given by IUPAC nomenclature. The naming of organic compound is done such that the structure of organic compound is correctly interpreted from the name.
Rules for writing IUPAC name from structural formula are
1. First identify the longest carbon chain.
2. The next step is to identify the groups attached to the longest chain.
3. Identify the position, location, and number of the substituent bonded to the carbon chain.
4. Use prefix di, tri, tetra if same type of substituent is present.
5. Name the substituents in alphabetical order.
Answer to Problem 10.40P
The IUPAC name for the given compound is
Explanation of Solution
The given compound is,
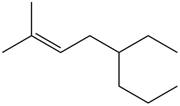
Figure 2
The numbering of carbon atoms present in longest chain of the given compound is shown below.
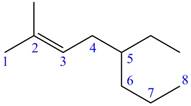
Figure 3
The longest carbon-carbon chain has eight carbon atoms. The root word used for eight carbon atoms is oct and the suffix used for
The IUPAC name for the given compound is
(c)
Interpretation: The IUPAC name for the given compound is to be stated.
Concept introduction: The systematic naming of organic compound is given by IUPAC nomenclature. The naming of organic compound is done such that the structure of organic compound is correctly interpreted from the name.
Rules for writing IUPAC name from structural formula are
1. First identify the longest carbon chain.
2. The next step is to identify the groups attached to the longest chain.
3. Identify the position, location, and number of the substituent bonded to the carbon chain.
4. Use prefix di, tri, tetra if same type of substituent is present.
5. Name the substituents in alphabetical order.
Answer to Problem 10.40P
The IUPAC name for the given compound is
Explanation of Solution
The given compound is,
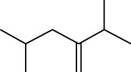
Figure 4
The numbering of carbon atoms present in longest chain of the given compound is shown below.
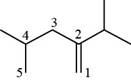
Figure 5
The longest carbon-carbon chain has five carbon atoms. The root word used for five carbon atoms is pent and the suffix used for
The IUPAC name for the given compound is
(d)
Interpretation: The IUPAC name for the given compound is to be stated.
Concept introduction: The systematic naming of organic compound is given by IUPAC. The naming of organic compound is done such that the structure of organic compound is correctly interpreted from the name.
Rules for writing IUPAC name from structural formula are
1. First identify the longest carbon chain.
2. The next step is to identify the groups attached to the longest chain.
3. Identify the position, location, and number of the substituent bonded to the carbon chain.
4. Use prefix di, tri, tetra if same type of substituent is present.
5. Name the substituents in alphabetical order.
Answer to Problem 10.40P
The IUPAC name for the given compound is
Explanation of Solution
The given compound is,
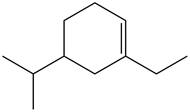
Figure 6
The numbering of carbon atoms present in longest carbon chain of the given compound is shown below.
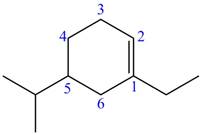
Figure 7
The longest carbon chain has six carbon atoms. The root word used for six carbon atoms is hex and the suffix used for
The IUPAC name for the given compound is
(e)
Interpretation: The IUPAC name for the given compound is to be stated.
Concept introduction: The systematic naming of organic compound is given by IUPAC nomenclature. The naming of organic compound is done such that the structure of organic compound is correctly interpreted from the name.
Rules for writing IUPAC name from structural formula are
1. First identify the longest carbon chain.
2. The next step is to identify the groups attached to the longest chain.
3. Identify the position, location, and number of the substituent bonded to the carbon chain.
4. Use prefix di, tri, tetra if same type of substituent is present.
5. Name the substituents in alphabetical order.
Answer to Problem 10.40P
The IUPAC name for the given compound is
Explanation of Solution
The given compound is,
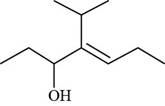
Figure 8
The numbering of carbon atoms present in longest carbon chain of the given compound is shown below.
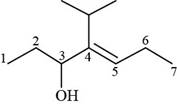
Figure 9
The longest carbon chain has seven carbon atoms. The root word used for seven carbon atoms is hept and the suffix used for
The IUPAC name for the given compound is
(f)
Interpretation: The IUPAC name for the given compound is to be stated.
Concept introduction: The systematic naming of organic compound is given by IUPAC. The naming of organic compound is done such that the structure of organic compound is correctly interpreted from the name.
Rules for writing IUPAC name from structural formula are
1. First identify the longest carbon chain.
2. The next step is to identify the groups attached to the longest chain.
3. Identify the position, location, and number of the substituent bonded to the carbon chain.
4. Use prefix di, tri, tetra if same type of substituent is present.
5. Name the substituents in alphabetical order.
Answer to Problem 10.40P
The IUPAC name for the given compound is
Explanation of Solution
The given compound is,
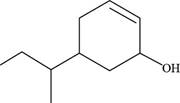
Figure 10
The numbering of carbon atoms present in longest carbon chain of the given compound is shown below.
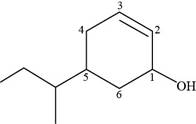
Figure 11
The longest carbon chain has six carbon atoms. The root word used for six carbon atoms is hex and the suffix used for
The IUPAC name of the given structure is
Want to see more full solutions like this?
Chapter 10 Solutions
Organic Chemistry - With Access (Custom)
- a. What is the chemical structure of biphenyl, circle functional groupsdifferent than alkane, alkene, alkyne? b. Is it polar or nonpolar? _______________________ c. What is its water solubility in g/L? __________________________arrow_forward3. a. What is the chemical structure of benzoic acid, circle functional groups different than alkane,alkene, alkyne? b. Is it polar or nonpolar? _______________________ c. What is its water solubility in g/L? __________________________arrow_forwardDraw the product formed when (CH3)2CHOH is treated with each reagent (a, b and c)arrow_forward
- What alkenes are formed when each alcohol is dehydrated with TsOH? Label the major product when a mixture resultsarrow_forwardGive the IUPAC or common name of each compound. (G- H)arrow_forwarda. What is the chemical structure of 2,6-dichloroindophenol, circle functional groups differentthan alkane, alkene, alkyne? b. Is it polar or nonpolar? _______________________ c. What is its water solubility in g/L? ___________arrow_forward
- 1.) What is the structure and IUPAC name of tert-butyl isopropyl acetylene? 2.) What is the structure of 2 methyl- dimethyl-alpha-hexylene? 2.) What is the structure of 3 methyl- dimethyl-beta-hexylene?arrow_forwardDraw the products formed when cis- and trans-but-2-ene are treated with CHCl3 and KOC(CH3)3arrow_forwardGive the structure corresponding to each IUPAC name. 2,4 dimethyl- 2 hexanolarrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
