
Concept explainers
Give the structure corresponding to each name.
a.
b.
c.
d.
(a)
Interpretation: The structure corresponding to the given
Concept introduction: The systematic naming of organic compound is given by IUPAC nomenclature. The naming of organic compound is done such that the structure of organic compound is correctly interpreted from the name.
Rules for writing structural formula from IUPAC are:
1. First identify the word root for the given compound.
2. The suffix used in the compound like –ene.
3. Identify the position, location, and number of the substituent bonded to the carbon chain.
The use of prefix
Answer to Problem 10.41P
The structure corresponding to
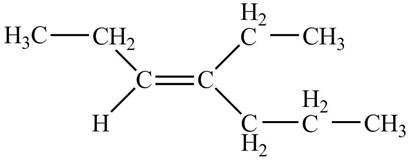
Explanation of Solution
The given IUPAC name is
Rules for writing structural formula from IUPAC are
1. First identify the word root for the given compound.
2. The suffix used in the compound like –ene.
3. Identify the position, location, and number of the substituent bonded to the carbon chain.
The use of prefix
The given name is
Thus, the correct structure of
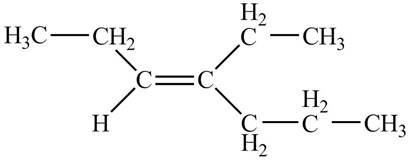
Figure 1
The structure corresponding to
(b)
Interpretation: The structure corresponding to the given
Concept introduction: The systematic naming of organic compound is given by IUPAC nomenclature. The naming of organic compound is done such that the structure of organic compound is correctly interpreted from the name.
Rules for writing structural formula from IUPAC are:
1. First identify the word root for the given compound.
2. The suffix used in the compound like –ene.
3. Identify the position, location, and number of the substituent bonded to the carbon chain.
Answer to Problem 10.41P
The structure corresponding to
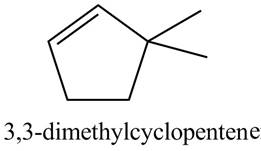
Explanation of Solution
Rules for writing structural formula from IUPAC are
1. First identify the word root for the given compound.
2. The suffix used in the compound like –ene.
3. Identify the position, location, and number of the substituent bonded to the carbon chain.
The given name is
Thus, the correct structure of
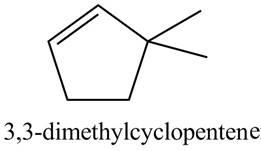
Figure 2
The structure corresponding to
(c)
Interpretation: The structure corresponding to
Concept introduction: The systematic naming of organic compound is given by IUPAC nomenclature. The naming of organic compound is done such that the structure of organic compound is correctly interpreted from the name.
Rules for writing structural formula from IUPAC are:
1. First identify the word root for the given compound.
2. The suffix used in the compound like –ene.
3. Identify the position, location, and number of the substituent bonded to the carbon chain.
Answer to Problem 10.41P
The structure corresponding to
Explanation of Solution
Rules for writing structural formula from IUPAC are
1. First identify the word root for the given compound.
2. The suffix used in the compound like –ene.
3. Identify the position, location, and number of the substituent bonded to the carbon chain.
The given name is
Thus, the correct structure of

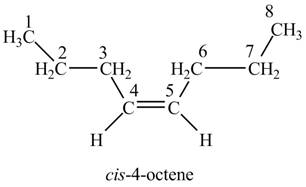
Figure 3
The structure corresponding to
(d)
Interpretation: The structure corresponding to
Concept introduction: The systematic naming of organic compound is given by IUPAC nomenclature. The naming of organic compound is done such that the structure of organic compound is correctly interpreted from the name.
Rules for writing structural formula from IUPAC are:
4. First identify the word root for the given compound.
5. The suffix used in the compound like –ene.
6. Identify the position, location, and number of the substituent bonded to the carbon chain.
Answer to Problem 10.41P
The structure corresponding to
Explanation of Solution
Rules for writing structural formula from IUPAC are
1. First identify the word root for the given compound.
2. The suffix used in the compound like –ene.
3. Identify the position, location, and number of the substituent bonded to the carbon chain.
The given name is
Thus, the correct structure of
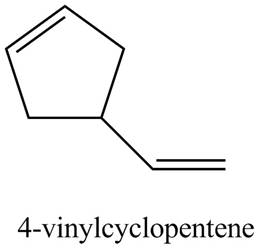
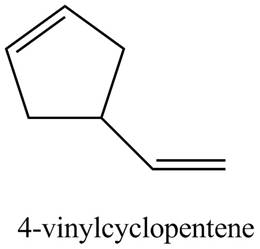
Figure 4
The structure corresponding to
(e)
Interpretation: The structure corresponding to
Concept introduction: The systematic naming of organic compound is given by IUPAC nomenclature. The naming of organic compound is done such that the structure of organic compound is correctly interpreted from the name.
Rules for writing structural formula from IUPAC are:
1. First identify the word root for the given compound.
2. The suffix used in the compound like –ene.
3. Identify the position, location, and number of the substituent bonded to the carbon chain.
The use of prefix
Answer to Problem 10.41P
The structure corresponding to
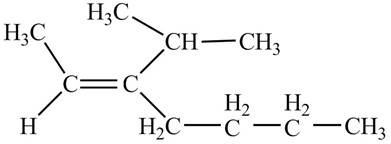
Explanation of Solution
Rules for writing structural formula from IUPAC are
1. First identify the word root for the given compound.
2. The suffix used in the compound like –ene.
3. Identify the position, location, and number of the substituent bonded to the carbon chain.
The use of prefix
The given name is
Thus, the correct structure of

Figure 5
The structure corresponding to
(f)
Interpretation: The structure corresponding to
Concept introduction: The systematic naming of organic compound is given by IUPAC nomenclature. The naming of organic compound is done such that the structure of organic compound is correctly interpreted from the name.
Rules for writing structural formula from IUPAC are:
1. First identify the word root for the given compound.
2. The suffix used in the compound like –ene.
3. Identify the position, location, and number of the substituent bonded to the carbon chain.
Answer to Problem 10.41P
The structure corresponding to
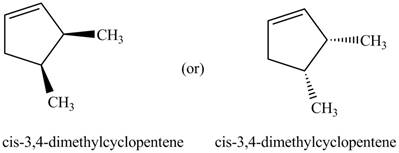
Explanation of Solution
Rules for writing structural formula from IUPAC are
1. First identify the word root for the given compound.
2. The suffix used in the compound like –ene.
3. Identify the position, location, and number of the substituent bonded to the carbon chain.
The given name is
Thus, the two correct structure of
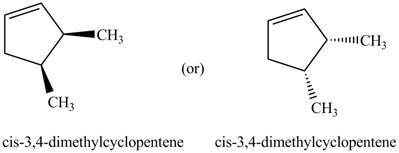
Figure 6
The structure corresponding to
(g)
Interpretation: The structure corresponding to
Concept introduction: The systematic naming of organic compound is given by IUPAC nomenclature. The naming of organic compound is done such that the structure of organic compound is correctly interpreted from the name.
Rules for writing structural formula from IUPAC are:
1. First identify the word root for the given compound.
2. The suffix used in the compound like –ene.
3. Identify the position, location, and number of the substituent bonded to the carbon chain.
Answer to Problem 10.41P
The structure corresponding to
![]()
Explanation of Solution
Rules for writing structural formula from IUPAC are
1. First identify the word root for the given compound.
2. The suffix used in the compound like –ene.
3. Identify the position, location, and number of the substituent bonded to the carbon chain.
The given name is
Thus, the correct structure of
![]()
Figure 7
The structure corresponding to
(h)
Interpretation: The structure corresponding to
Concept introduction: The systematic naming of organic compound is given by IUPAC nomenclature. The naming of organic compound is done such that the structure of organic compound is correctly interpreted from the name.
Rules for writing structural formula from IUPAC are:
1. First identify the word root for the given compound.
2. The suffix used in the compound like –ene.
3. Identify the position, location, and number of the substituent bonded to the carbon chain.
Answer to Problem 10.41P
The structure corresponding to
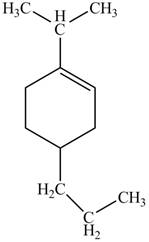
Explanation of Solution
Rules for writing structural formula from IUPAC are
1. First identify the word root for the given compound.
2. The suffix used in the compound like –ene.
3. Identify the position, location, and number of the substituent bonded to the carbon chain.
The given name is
Thus, the correct structure of
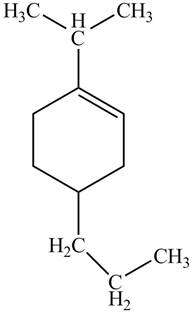
Figure 8
The structure corresponding to
Want to see more full solutions like this?
Chapter 10 Solutions
Connect Access Card For Organic Chemistry
- how can i rank these isomers from lowest bp to highest please? Trans-pent-2-ene Cis-pent-2-ene 3-methylbut-1-ene 2-methylbut-1-ene 2-methylbut-2-ene Cyclopentane Methylcyclobutane Ethylcyclopropane 1,2-dimethylcyclopropane 1,1-dimethylcyclopropane (don't answer only, need explaination)arrow_forwardhow can i rank these isomers from lowest bp to highest please? Trans-pent-2-ene Cis-pent-2-ene 3-methylbut-1-ene 2-methylbut-1-ene 2-methylbut-2-ene Cyclopentane Methylcyclobutane Ethylcyclopropane 1,2-dimethylcyclopropane 1,1-dimethylcyclopropanearrow_forwardRank the following groups in order of decreasing priority. a.−COOH, −H, −NH2, −OH b.−H, −CH3, −Cl, −CH2Cl c. −CH2CH3, −CH3, −H, −CH(CH3)2 d.−CH=CH2, −CH3, −C≡CH, −Harrow_forward
- Show all steps and reagents needed to convert cyclohexane into each compound: (a) the two enantiomers of trans-1,2- dibromocyclohexane; and (b) 1,2-epoxycyclohexane.arrow_forwardCompound X is insoluble in cold KMnO4, Br2 in CCl4, and conc. H2SO4. Compound X is most likely: a. an alkane b. none of these c. an alkene d. an alcohol e. an alkyl halide Indicate which of the ff. statements regarding nucleophilicity is incorrect. F- is more nucleophilic, hence, more reactive towards methyl iodide than Cl-. Second row elements are more nucleophilic than first row elements of comparable basicity. The rate of SN2 reaction may be markedly affected by the nucleophilicity of the attacking atom. Nucleophilicity is the affinity of a nucleophile to an electrophilic carbon Which of the following alkynes can be deprotonated by NaNH2 in liquid NH3? 3-Methylhex-2-yne Pent-2-yne 3-Methylbutyne none of these Hex-3-ynearrow_forward1. Using Br2 in C2H4Br2 will result in HBr and ______. a. C2H3Cl3 b. C2H4Cl3 c. C2H2Cl3 d. none of the above 2. How many halogenation are posible in propane? a. 3 b. 8 c. 6 d. 10 3.Sulfonation of pentane will result in ________ and water. a. C5H11SO3H b. C5H12SO3H c. C5H14SO3H d. none of the above 4.Nitration of hexane will result in ________ and water. a. C6H13SO3H b. C6H15NO2 c. C6H13NO2 d. C6H14NO2 5.How many moles of O2 in heating a C12H26 (dodecane) a. 27 b. 37 c. 24 d. none of the abovearrow_forward
- Conversion of Compound into complete structure. A.CH3(CH2)8CH3 B. CH3(CH2)4OH C. CH3CCl3 D. CH3(CH2)4CH(CH3)2 E. (CH3)2CHCH2NH2arrow_forwardRank the following groups in order of decreasing priority. −Cl, −CH3, −SH, −OHarrow_forwardIndicate whether each statement is true or false. (a) Butanecontains carbons that are sp2 hybridized. (b) Cyclohexaneis another name for benzene. (c) The isopropyl group containsthree sp3-hybridized carbons. (d) Olefin is anothername for alkyne.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

