
Concept explainers
Interpretation:
A mechanism for the given conversion has to be shown.
Concept Introduction:
Acid-catalyzed dehydration of alcohols: An alcohol can be converted into an
Ease of dehydration of alcohols:
The ease of acid-catalyzed dehydration of alcohols is in the order as follows,
Ozonolysis: The unsaturated bonds of alkene,
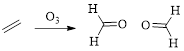
Want to see the full answer?
Check out a sample textbook solution
Chapter 10 Solutions
ORGANIC CHEMISTRY-OWL V2 ACCESS
- Classify the following reactions as addition (A), substitution (S), elimination (E) or rearrangement (R).arrow_forwardWhat is/are the product(s) formed when K2Cr2O7? H,C- QHU C- CH, H H CH is oxidized with H CH CH H,C- CH H. CH3 then CH -CH2 CH H CH CH3(CH2)4COOH H3C- ར་ ཨ་ ཨ་་arrow_forwardi need the answer quicklyarrow_forward
- Describe a sequence of reactions by which cis-2-pentene could be prepared from acetylene.arrow_forwardв. Odoriferous Organic Compounds Below each company identify the source or use as recognized by familiar odor. CH3 c-CH-CH,CH,c=CHCH,OH CH3 CH, C-CH-CH,CH,-C-CH-C CH3 CH CH3 H. Geraniol Citral CH 3 С -Н CH3 CH3- CH OCH3 он CH3 CH3 Pinene Carvone Vanillin CH он CH OCH3 CHC-CH ČH,CH-CH2 CH CH3 CH, Menthol Camphor Eugenol C1 co,CH, OH CH=CH-C Methyl salicylate Cinnamaldehyde p-Dichlorobenzenearrow_forwardCompound X (structure shown below) has a molecular formula C5H1o and reacts with H2/Pt to give compound Y, C5H12. What is the name of the reaction involved to produce Compound Y? H2C H3C CH3 Hydration Hydrogenation Halogenation Addition of halohydrinarrow_forward
- Show how to convert ethylene to 1,2-Dibromoethane compound.arrow_forwardEither the reactant (X) or the major organic product is missing from the equation below. Draw the missing compound. CH3 O || CH3CH2ĊCH,ČOCH3 Pd/C X + H2 ČH3arrow_forward2. Provide the reactant, reagents, or products for the following reactions. Br NH₂ $ Phthalimide NaOH, Br₂ H₂O Ag₂O H₂O, Aarrow_forward
- Explain why alkenes undergo electrophilic addition reactionsand alkanes do not. Use Butane, But-2-ene and chlorine asexamples. Clearly show the reaction mechanism.arrow_forwardIn an advanced synthetic chemistry experiment, a researcher prepares a compound, ZY-7, by reacting a ketone (C5H100) with hydroxylamine (NH2OH), followed by heating in the presence of an acid catalyst. The resulting compound, ZY-7, is then treated with a solution of sodium nitrite (NaNO2) and hydrochloric acid (HCI) at low temperature. Identify the class of compound that ZY-7 most likely belongs to after this series of reactions." A) Amide B) Oxime C) Nitro compound D) Diazonium salt E) Ester Don't use chatgpt please provide valuable answerarrow_forwardMethyl isocyanate, CH3 -N= C = O, is used in the industrial synthesis of a type of pesticide and herbicide known as a carbamate. As a historical note, an industrial accident in Bhopal, India, in 1984 resulted in leakage of an unknown quantity of this chemical into the air. An estimated 200,000 people were exposed to its vapors, and over 2000 of these people died. Q.) Methyl isocyanate reacts with strong acids, such as sulfuric acid, to form a cation. Will this molecule undergo protonation more readily on its oxygen or nitrogen atom? In considering contributing structures to each hybrid, do not consider structures in which more than one atom has an incomplete octetarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





