
Concept explainers
(a)
Interpretation:
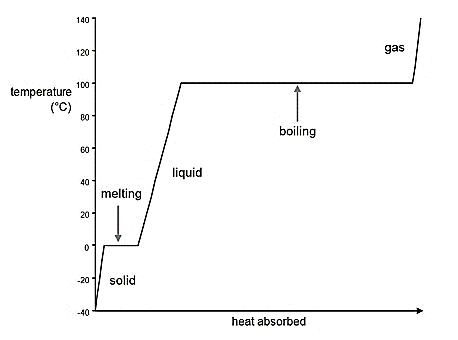
The heating curve for ethanol from -150°C to 100°C should be drawn.
Concept Introduction:
When the system receives heat, energy is transferred into the system. In result, to the energy received by the system, the change in it occurs. For an example: By increase in temperature of the system.
A graph which represents the phase changes of the substance occurs with decrease or increase in temperature is known as heating curve.
General representation of graph is given by:
(b)
Interpretation:
The required energy in kilojoules should be calculated.
Concept Introduction:
The mathematical expression for energy required is given by:
Where, q = amount of energy required
m= mass of substance
(c)
Interpretation:
The volume of ethanol in liters should be calculated.
Concept Introduction:
An arithmetical multiplier which is used for converting a quantity expressed in one unit into another equivalent set of units is said to be conversion factor.
The conversion factor for gallon into liters is given by:
Since, 1 gallon = 3.785 L
Thus,
(d)
Interpretation:
The balanced chemical equation for complete combustion of ethanol should be written.
Concept Introduction:
A
(e)
Interpretation:
The mass of carbon dioxide in kilograms which is produced by the complete combustion of ethanol in 15.0-gal gas tank should be calculated.
Concept Introduction:
Density is defined as the ratio of mass to the volume of a substance.
The mathematical expression of density is given by:
(f)
Interpretation:
The intermolecular forces (strong) present between the liquid molecules of ethanol should be determined.
Concept Introduction:
An intermolecular (forces present between the molecule) or intramolecular (forces present within the molecule) interaction which takes place between molecules with hydrogen bond donors and molecules with hydrogen bond acceptors is known as hydrogen bonding. When the hydrogen is linked to an electronegative atom is known as hydrogen bond donors whereas when a lone pair of electrons is present on electronegative atom in a molecule is known as hydrogen bond acceptors. It is stronger than van der Waals forces but less strong than ionic and covalent bonds.
Trending nowThis is a popular solution!

Chapter 10 Solutions
Basic Chemistry, Books a la Carte Edition (5th Edition)
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





