
Concept explainers
Interpretation:
The empirical formula for the given compound should be predicted.
Concept introduction:
The representation of atoms of a compound in simple whole number ratio is known as empirical formula.
The rules for determining empirical formula is given as follows:
- Determine the mass of elements in given compound.
- Calculate number of moles using molar mass of each compound or element.
- Divide each number of moles by smallest number of mole value calculated in second step.
- Round the value calculated in step 3 to nearest whole number.
Answer to Problem 6STP
The empirical formula for given compound is
Explanation of Solution
Assuming the percentage of the elements is given mass by mass which means specific amount of the element is present in 100 g of the solution.
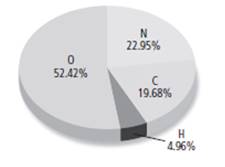
1. Mass of carbon = 19.68 g
Mass of hydrogen = 4.96 g
Mass of oxygen = 52.42 g
Mass of nitrogen = 22.95 g
2. Now, calculating the moles of carbon, hydrogen, nitrogen and oxygen using their molar mass as follows:
3. Now, dividing each mole value by smallest mole value as follows:
Thus, the empirical formula of given compound is
Hence, the correct option is (C) that is
Chapter 10 Solutions
Glencoe Chemistry: Matter and Change, Student Edition
Additional Science Textbook Solutions
Chemistry: Structure and Properties (2nd Edition)
Chemistry: Structure and Properties
CHEMISTRY-TEXT
Introductory Chemistry (6th Edition)
Chemistry: The Central Science (14th Edition)
General Chemistry: Principles and Modern Applications (11th Edition)
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





