
CHEMISTRY: ATOMS FIRST VOL 1 W/CONNECT
14th Edition
ISBN: 9781259327933
Author: Burdge
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 10.2, Problem 1PPC
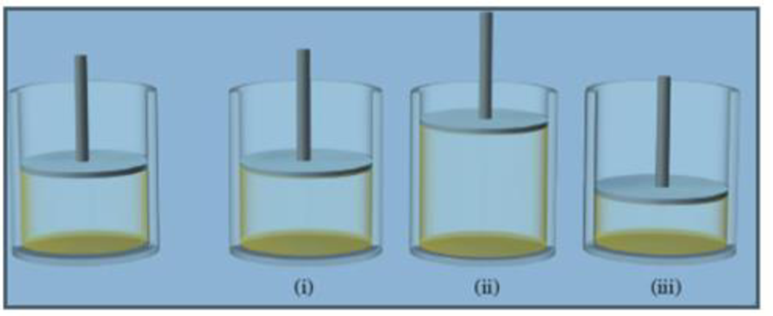
The diagram on the left shows a system before a process. Which of the diagrams on the right could represent the system after it undergoes a process in which the system absorbs heat and ΔU is positive?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Chapter 10 Solutions
CHEMISTRY: ATOMS FIRST VOL 1 W/CONNECT
Ch. 10.1 - Prob. 10.1.1SRCh. 10.1 - Prob. 10.1.2SRCh. 10.1 - Prob. 10.1.3SRCh. 10.2 - Calculate the overall change in internal energy,...Ch. 10.2 - Calculate the change in total internal energy for...Ch. 10.2 - Calculate the magnitude of q for a system that...Ch. 10.2 - The diagram on the left shows a system before a...Ch. 10.2 - Prob. 10.2.1SRCh. 10.2 - Prob. 10.2.2SRCh. 10.3 - Determine the work done (in joules) when a sample...
Ch. 10.3 - Calculate the work done by or on the system during...Ch. 10.3 - (a) Against what external pressure must a gas...Ch. 10.3 - The diagram on the left shows a sample of gas...Ch. 10.3 - Given the thermochemical equation for...Ch. 10.3 - Calculate the solar energy required to produce...Ch. 10.3 - Prob. 3PPBCh. 10.3 - The diagrams represent systems before and after...Ch. 10.3 - Prob. 10.3.1SRCh. 10.3 - Prob. 10.3.2SRCh. 10.4 - Prob. 10.4WECh. 10.4 - Prob. 4PPACh. 10.4 - Prob. 4PPBCh. 10.4 - A metal pellet with a mass of 100.0 g. originally...Ch. 10.4 - What would the final temperature be if the pellet...Ch. 10.4 - Prob. 5PPBCh. 10.4 - Prob. 5PPCCh. 10.4 - A Famous Amos bite-sized chocolate chip cookie...Ch. 10.4 - A serving of Grape-Nuts cereal (5.80 g) is burned...Ch. 10.4 - Prob. 6PPBCh. 10.4 - Suppose an experiment to determine the energy...Ch. 10.4 - Prob. 10.4.1SRCh. 10.4 - Prob. 10.4.2SRCh. 10.4 - Prob. 10.4.3SRCh. 10.4 - Prob. 10.4.4SRCh. 10.5 - Given the following thermochemical equations....Ch. 10.5 - Use the thermochemical equations provided in...Ch. 10.5 - Prob. 10.5.1SRCh. 10.5 - Prob. 10.5.2SRCh. 10.6 - Prob. 10.8WECh. 10.6 - Using data from Appendix 2, calculate Hrn for...Ch. 10.6 - Prob. 8PPBCh. 10.6 - The diagrams represent a system before and after a...Ch. 10.6 - Given the following information, calculate the...Ch. 10.6 - Use the following data to calculate Hf for...Ch. 10.6 - Prob. 9PPBCh. 10.6 - The diagrams represent a system before and after a...Ch. 10.6 - Prob. 10.6.1SRCh. 10.6 - Prob. 10.6.2SRCh. 10.6 - Prob. 10.6.3SRCh. 10.7 - Use bond enthalpies from Table 10.4 to estimate...Ch. 10.7 - Use bond enthalpies from fable 10.4 to estimate...Ch. 10.7 - Prob. 10PPBCh. 10.7 - Prob. 10PPCCh. 10.7 - Prob. 10.7.1SRCh. 10.7 - Prob. 10.7.2SRCh. 10.7 - Prob. 10.7.3SRCh. 10.7 - Prob. 10.7.4SRCh. 10.8 - Prob. 10.11WECh. 10.8 - Prob. 11PPACh. 10.8 - The lattice energy of MgO is 3890 kJ/mol, and the...Ch. 10.8 - Prob. 11PPCCh. 10.8 - Prob. 10.8.1SRCh. 10.8 - Prob. 10.8.2SRCh. 10 - Define these terms: system, surroundings, thermal...Ch. 10 - What is heat? How does heat differ from thermal...Ch. 10 - Prob. 10.3QPCh. 10 - Define these terms: thermochemistry, exothermic...Ch. 10 - Prob. 10.5QPCh. 10 - Describe two exothermic processes and two...Ch. 10 - Decomposition reactions are usually endothermic,...Ch. 10 - On what law is the first law of thermodynamics...Ch. 10 - Explain what is meant by a state function. Give...Ch. 10 - Prob. 10.10QPCh. 10 - Prob. 10.11QPCh. 10 - Prob. 10.12QPCh. 10 - Prob. 10.13QPCh. 10 - Prob. 10.14QPCh. 10 - Prob. 10.15QPCh. 10 - Prob. 10.16QPCh. 10 - Define these terms: enthalpy and enthalpy of...Ch. 10 - Prob. 10.18QPCh. 10 - Prob. 10.19QPCh. 10 - Prob. 10.20QPCh. 10 - Prob. 10.21QPCh. 10 - A gas expands and does PV work on the surroundings...Ch. 10 - Prob. 10.23QPCh. 10 - Prob. 10.24QPCh. 10 - Consider the reaction at a certain temperature. If...Ch. 10 - Prob. 10.26QPCh. 10 - Prob. 10.27QPCh. 10 - Prob. 10.28QPCh. 10 - Prob. 10.1VCCh. 10 - Prob. 10.2VCCh. 10 - Prob. 10.3VCCh. 10 - Prob. 10.4VCCh. 10 - Prob. 10.5VCCh. 10 - Prob. 10.6VCCh. 10 - Prob. 10.7VCCh. 10 - Referring to the process depicted in Figure 10.10,...Ch. 10 - What is the difference between specific heat and...Ch. 10 - Define calorimetry and describe two commonly used...Ch. 10 - Prob. 10.31QPCh. 10 - Prob. 10.32QPCh. 10 - A sheet of gold weighing 10.0 g and at a...Ch. 10 - Prob. 10.34QPCh. 10 - A quantity of 2.00 102 mL of 0.862 M HC1 is mixed...Ch. 10 - Prob. 10.36QPCh. 10 - Prob. 10.37QPCh. 10 - Prob. 10.38QPCh. 10 - A 25.95-g sample of methanol at 35.6C is added to...Ch. 10 - Prob. 10.40QPCh. 10 - Prob. 10.41QPCh. 10 - Prob. 10.42QPCh. 10 - Prob. 10.43QPCh. 10 - Prob. 10.44QPCh. 10 - Prob. 10.45QPCh. 10 - Prob. 10.46QPCh. 10 - Prob. 10.47QPCh. 10 - Prob. 10.48QPCh. 10 - Prob. 10.49QPCh. 10 - Prob. 10.50QPCh. 10 - What is meant by the standard-state condition?Ch. 10 - How are the standard enthalpies of an element and...Ch. 10 - What is meant by the standard enthalpy of a...Ch. 10 - Write the equation for calculating the enthalpy of...Ch. 10 - Prob. 10.55QPCh. 10 - Prob. 10.56QPCh. 10 - Prob. 10.57QPCh. 10 - Calculate the heats of combustion for the...Ch. 10 - Calculate the heats of combustion for the...Ch. 10 - Prob. 10.60QPCh. 10 - Prob. 10.61QPCh. 10 - Prob. 10.62QPCh. 10 - From the standard enthalpies of formation,...Ch. 10 - Prob. 10.64QPCh. 10 - Prob. 10.65QPCh. 10 - Prob. 10.66QPCh. 10 - Which is the more negative quantity at 25C: Hf for...Ch. 10 - Prob. 10.68QPCh. 10 - Prob. 10.69QPCh. 10 - Prob. 10.70QPCh. 10 - Prob. 10.71QPCh. 10 - Prob. 10.72QPCh. 10 - Prob. 10.73QPCh. 10 - Prob. 10.74QPCh. 10 - Prob. 10.75QPCh. 10 - Prob. 10.76QPCh. 10 - For the reaction 2C2H6(g)+7O2(g)4CO2(g)+6H2O(g)...Ch. 10 - Prob. 10.78QPCh. 10 - Prob. 10.79QPCh. 10 - Prob. 10.9VCCh. 10 - Prob. 10.10VCCh. 10 - Prob. 10.11VCCh. 10 - Prob. 10.12VCCh. 10 - Explain how the lattice energy of an ionic...Ch. 10 - Specify which compound in each of the following...Ch. 10 - Prob. 10.82QPCh. 10 - Prob. 10.83QPCh. 10 - Prob. 10.84QPCh. 10 - Prob. 10.85QPCh. 10 - Prob. 10.86QPCh. 10 - Prob. 10.87QPCh. 10 - Hydrazine (N2H4) decomposes according to the...Ch. 10 - Prob. 10.89QPCh. 10 - Prob. 10.90QPCh. 10 - Prob. 10.91QPCh. 10 - Prob. 10.92QPCh. 10 - Prob. 10.93QPCh. 10 - Prob. 10.94QPCh. 10 - You are given the following data....Ch. 10 - Prob. 10.96QPCh. 10 - Prob. 10.97QPCh. 10 - Prob. 10.98QPCh. 10 - Prob. 10.99QPCh. 10 - Compare the heat produced by the complete...Ch. 10 - The so-called hydrogen economy is based on...Ch. 10 - Prob. 10.102QPCh. 10 - Prob. 10.103QPCh. 10 - Prob. 10.104QPCh. 10 - Prob. 10.105QPCh. 10 - Prob. 10.106QPCh. 10 - Prob. 10.107QPCh. 10 - Prob. 10.108QPCh. 10 - A certain gas initially at 0.050 L undergoes...Ch. 10 - Prob. 10.110QPCh. 10 - The first step in the industrial recovery of zinc...Ch. 10 - Calculate the standard enthalpy change for the...Ch. 10 - Portable hot packs are available for skiers and...Ch. 10 - Prob. 10.114QPCh. 10 - Prob. 10.115QPCh. 10 - Prob. 10.116QPCh. 10 - Prob. 10.117QPCh. 10 - Prob. 10.118QPCh. 10 - Prob. 10.119QPCh. 10 - Prob. 10.120QPCh. 10 - Prob. 10.121QPCh. 10 - Prob. 10.122QPCh. 10 - Prob. 10.123QPCh. 10 - Prob. 10.124QPCh. 10 - Prob. 10.125QPCh. 10 - Vinyl chloride (C2H3Cl) differs from ethylene...Ch. 10 - Prob. 10.127QPCh. 10 - Prob. 10.128QPCh. 10 - Prob. 10.129QPCh. 10 - Determine the standard enthalpy of formation of...Ch. 10 - Prob. 10.131QPCh. 10 - Prob. 10.132QPCh. 10 - Prob. 10.133QPCh. 10 - Prob. 10.134QPCh. 10 - Prob. 10.135QPCh. 10 - Prob. 10.136QPCh. 10 - Both glucose and fructose arc simple sugars with...Ch. 10 - About 6.0 1013 kg of CO2 is fixed (converted to...Ch. 10 - Experiments show that it takes 1656 kJ/mol to...Ch. 10 - From a thermochemical point of view, explain why a...Ch. 10 - Prob. 10.141QPCh. 10 - Prob. 10.142QPCh. 10 - Prob. 10.143QPCh. 10 - Prob. 10.144QPCh. 10 - Prob. 10.145QPCh. 10 - Prob. 10.146QPCh. 10 - Prob. 10.147QPCh. 10 - Prob. 10.148QPCh. 10 - A drivers manual states that the stopping distance...Ch. 10 - Prob. 10.150QPCh. 10 - Prob. 10.151QPCh. 10 - Prob. 10.152QPCh. 10 - When 1.034 g of naphthalene (C10H8), is burned in...Ch. 10 - Prob. 10.154QPCh. 10 - A gas company in Massachusetts charges 27 cents...Ch. 10 - Prob. 10.156QPCh. 10 - Prob. 10.157QPCh. 10 - According to information obtained from...Ch. 10 - Using data from Appendix 2, calculate the standard...Ch. 10 - Using data from Appendix 2, calculate the standard...Ch. 10 - Prob. 10.3KSPCh. 10 - Prob. 10.4KSP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Explain the difference between heat capacity and specific heat of a substance.arrow_forwardA 50-mL solution of a dilute AgNO3 solution is added to 100 mL of a base solution in a coffee-cup calorimeter. As Ag2O(s) precipitates, the temperature of the solution increases from 23.78 C to 25.19 C. Assuming that the mixture has the same specific heat as water and a mass of 150 g, calculate the heat q. Is the precipitation reaction exothermic or endothermic?arrow_forwardAlloys When a 58.8-g piece of hot alloy is placed in125 g of cold water in a calorimeter, the temperature ofthe alloy decreases by 106.1°C, while the temperature ofthe water increases by 10.5°C. What is the specific heat ofthe alloy?arrow_forward
- Another reaction that is used to propel rockets is N2O4(l)+2N2H4(l)3N2(g)+4H2O(g) This reaction has the advantage that neither product is toxic, so no dangerous pollution is released. When the reaction consumes 10.0 g liquid N2O4, it releases 124 kJ of heat. (a) Is the sign of the enthalpy change positive or negative? (b) What is the value of H for the chemical equation if it is understood to be written in molar quantities?arrow_forwardMetallurgy A 25.0-g bolt made of an alloy absorbed250 J of heat as its temperature changed from 25.0°C to78.0°C. What is the specific heat of the alloy?arrow_forwardThe temperature of the cooling water as it leaves the hot engine of an automobile is 240 F. After it passes through the radiator it has a temperature of 175 F. Calculate the amount of heat transferred from the engine to the surroundings by one gallon of water with a specific heat of 4.184 J/g oC.arrow_forward
- 9.41 Under what conditions does the enthalpy change equal the heat of a process?arrow_forwardThe process of dissolving ammonium nitrate, NH4NO3, in water is an endothermic process. What is the sign of q? If you were to add some ammonium nitrate to water in a flask, would you expect the flask to feel warm or cool?arrow_forwardA sample of nickel is heated to 99.8C and placed in a coffee-cup calorimeter containing 150.0 g water at 23.5C. After the metal cools, the final temperature of metal and water mixture is 25.0C. If the specific heat capacity of nickel is 0.444 J/C g, what mass of nickel was originally heated? Assume no heat loss to the surroundings.arrow_forward
- A soluble salt, MX2, is added to water in a beaker. The equation for the dissolving of the salt is: MX2(s)M2+(aq)+2X(aq);H0 a Immediately after the salt dissolves, is the solution warmer or colder? b Indicate the direction of heat flow, in or out of the beaker, while the salt dissolves. c After the salt dissolves and the water returns to room temperature, what is the value of q for the system?arrow_forwardDetermine whether the statements given below are true or false. Consider an endothermic process taking place in a beaker at room temperature. (a) Heat flows from the surroundings to the system. (b) The beaker is cold to the touch. (c) The pressure of the system decreases. (d) The value of q for the system is positive.arrow_forwardWhen solid iron burns in oxygen gas (at constant pressure) to produce Fe2O3(s), 1651 kJ of heat is released for every 4 mol of iron burned. How much heat is released when 10.3 g Fe2O3(s) is produced (at constant pressure)? What additional information would you need to calculate the heat released to produce this much Fe2O3(s) if you burned iron in ozone gas, O3(g), instead of O2(g)?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning,
Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning, Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Living By Chemistry: First Edition Textbook
Chemistry
ISBN:9781559539418
Author:Angelica Stacy
Publisher:MAC HIGHER

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Physical Chemistry
Chemistry
ISBN:9781133958437
Author:Ball, David W. (david Warren), BAER, Tomas
Publisher:Wadsworth Cengage Learning,

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning
The Laws of Thermodynamics, Entropy, and Gibbs Free Energy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=8N1BxHgsoOw;License: Standard YouTube License, CC-BY