
Essential Organic Chemistry, Global Edition
3rd Edition
ISBN: 9781292089034
Author: Paula Yurkanis Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 10.28, Problem 46P
Describe the 1H NMR spectrum you would expect for each of the following compounds, Indicating the relative positions of the signals:
- a. BrCH2CH2CH2CH2Br
- b. CH3OCH2CH2CH2Br
- c. CH3CH2OCH2CH3
- d. CH3CH2OCH2Cl
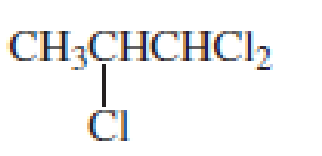
- e. CH3OCH2CH2CH2OCH3
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
For each of the molecules in Problem 16.39, determine how many signals should appear in its 13C NMR spectrum.
1) Write the peak number and peak ratio by drawing the NMR spectrum of the compounds given belowa) CH3CHBrCH3b) CH3CH2CH2I
The 1H NMR spectrum of N,N-dimethylformamide shows three singlets at2.9, 3.0, and 8.0 ppm. Explain why the two CH3 groups are not equivalentto each other, thus giving rise to two NMR signals.
Chapter 10 Solutions
Essential Organic Chemistry, Global Edition
Ch. 10.1 - Prob. 1PCh. 10.2 - What would distinguish the mass spectrum of...Ch. 10.2 - Prob. 3PCh. 10.3 - Prob. 5PCh. 10.3 - Suggest possible molecular formulas for a compound...Ch. 10.3 - Prob. 7PCh. 10.4 - Prob. 8PCh. 10.4 - Prob. 9PCh. 10.5 - Prob. 10PCh. 10.5 - Prob. 11P
Ch. 10.6 - Identify the ketone responsible for the mass...Ch. 10.6 - Prob. 13PCh. 10.8 - Prob. 14PCh. 10.8 - Prob. 15PCh. 10.12 - Which will occur at a larger wavenumber: a. a C :...Ch. 10.13 - Which will occur at a larger wavenumber: a. the C...Ch. 10.13 - Prob. 18PCh. 10.13 - Prob. 19PCh. 10.13 - Which will show an O 8 H stretch at a larger...Ch. 10.14 - Prob. 21PCh. 10.14 - Prob. 22PCh. 10.15 - Prob. 23PCh. 10.15 - Prob. 24PCh. 10.17 - Prob. 25PCh. 10.18 - Prob. 26PCh. 10.18 - Prob. 27PCh. 10.19 - Prob. 28PCh. 10.19 - Prob. 29PCh. 10.22 - How many signals would you expect to see in the 1H...Ch. 10.22 - Prob. 31PCh. 10.22 - Prob. 32PCh. 10.23 - Where would you expect to find the 1H NMR signal...Ch. 10.24 - Prob. 34PCh. 10.25 - Prob. 35PCh. 10.25 - Prob. 36PCh. 10.25 - Prob. 37PCh. 10.26 - Prob. 38PCh. 10.26 - Which of the following compounds is responsible...Ch. 10.27 - Prob. 40PCh. 10.27 - Prob. 41PCh. 10.27 - The 1H NMR spectra of two carboxylic acids with...Ch. 10.28 - Prob. 43PCh. 10.28 - Prob. 44PCh. 10.28 - Prob. 45PCh. 10.28 - Describe the 1H NMR spectrum you would expect for...Ch. 10.28 - Identify the compound with molecular formula...Ch. 10.29 - Prob. 48PCh. 10.29 - Prob. 49PCh. 10.29 - Identify the compound with a molecular formula of...Ch. 10 - In the mass spectrum of the following compounds,...Ch. 10 - For each of the following pairs of compounds,...Ch. 10 - Draw the structure of a saturated hydrocarbon that...Ch. 10 - Prob. 54PCh. 10 - Prob. 55PCh. 10 - How could you use UV spectroscopy to distinguish...Ch. 10 - Prob. 57PCh. 10 - Predict the relative intensities of the molecular...Ch. 10 - Prob. 59PCh. 10 - List the following compounds in order from highest...Ch. 10 - How can 1H NMR be used to prove that the addition...Ch. 10 - There are four esters with molecular formula...Ch. 10 - Prob. 63PCh. 10 - Prob. 64PCh. 10 - Each of the IR spectra presented here is...Ch. 10 - Prob. 66PCh. 10 - Five compounds are shown for each of the following...Ch. 10 - Prob. 68PCh. 10 - Prob. 69PCh. 10 - Phenolphthalein is an acidbase indicator. In...Ch. 10 - Which one of the following five compounds produced...Ch. 10 - Prob. 72PCh. 10 - Prob. 73PCh. 10 - Prob. 74PCh. 10 - How could 1H NMR distinguish between the compounds...Ch. 10 - Prob. 76PCh. 10 - Prob. 77PCh. 10 - The 1H NMR spectra of three isomers with molecular...Ch. 10 - The 1H NMR spectra of three isomers with molecular...Ch. 10 - Identify the following compounds. (Relative...Ch. 10 - An alkyl halide reacts with an alkoxide ion to...Ch. 10 - Determine the structure of a compound with...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Ascaridole is a natural product that has been used to treat intestinal worms. Explain why the two methyls on the isopropyl group in ascaridole appear in its 1H-NMR spectrum as four lines of equal intensity, with two sets of two each separated by 7 Hz.arrow_forwardAssign the peaks in the 1H NMR spectrum of eugenol (Fig. 6.23) to specific protons in the molecule. The OH peak is at 5.1 ppm.arrow_forwardThe IR and proton NMR of compound C12H18 shown below. A) what is it U value? B) How many equivalent H’s per molecule are responsible for singlet peak at 1.35 ppm in the proton NMR C) How many equivalent H’s per molecule are responsible for singlet peak at 2.35 ppm D) Draw the structure base on the Ir and NMrarrow_forward
- 03) Below are presented two 1H NMR spectra. Among the compounds pentan-3-ol, pentan2-ol, pent-3-amine, pent-2-amine, pentan-2-one, which one corresponds to spectrum A and which one to B? Justify based on chemical shifts and multiplicity of spectra signalsarrow_forwardWhich of these molecules is the best IR spectrum A. 3- Phenypropanoic acid B. 2-Nethy1-2 - penatanol C none of these D. Propiophenonearrow_forwardAnswer the following questions about each of the hydroxy ketones: 1-hydroxybutan-2-one (A) and 4-hydroxybutan-2-one (B). a.) How many signals are observed in the 1H NMR spectrum?b.) Give the splitting observed for each type of proton as well as its approximate chemical shift.arrow_forward
- 1. Consider the IR and MS spectra of a partially unknown compound. a) Suggest a fragmentation pattern to explain the formation of the base peak. b) Determine the structure of the compound. Indicate how the MS and IR spectra supports your suggested structure.arrow_forwardDetermine the correct structure of the compound, which shows following 13C NMR DEPT-135 data is 13C NMR DEPT-135: negative peaks at δ30.2, 31.9, 61.8, 114.7 ppm; positive peak at 130.4 ppm.arrow_forwardThe 1H NMR spectrum of N,N-dimethylformamide shows three singlets at 2.9, 3.0, and 8.0 ppm. Explain why the two CH3 groups are not equivalent to each other, thus giving rise to two NMR signals.arrow_forward
- Draw the structure for the two compounds below and give the number of peaks that would be observed in their 1H and 13C NMR spectrums. m-chlorobenzamide s-cis-2,3-dimethyl-1,3-butadiene I appreciate the help!arrow_forwardA quartet was observed in the 1H NMR spectrum of an unknown organic compound. Which of the following is the least likely the structure of the unknown compound? CH3CH2CH2CH2Br BrCH2CH2CH2Br BrCH2CH2CH3 CH3CH2CH2CH3 CH3CH2CH(Br)CH3arrow_forwardA. Predict the splitting pattern of the compound (a) in the 1H1H NMR spectrum. B. Predict the splitting pattern of the compound (b) in the 1H1H NMR spectrum. C. Predict the splitting pattern of the compound (c) in the 1H1H NMR spectrum. D. Predict the splitting pattern of the compound (d) in the 1H1H NMR spectrum.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:9781305577190
Author:Kenneth L. Williamson, Katherine M. Masters
Publisher:Brooks Cole

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
NMR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=SBir5wUS3Bo;License: Standard YouTube License, CC-BY