
Concept explainers
a)
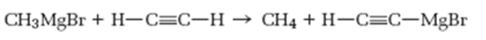
Interpretation:
As a base how much strong Grignard reagent is to be stated. Further based on the pKa values of
Concept introduction:
Stronger acids have low pKa values. A strong acid will yield a weak conjugate base. Similarly Weak acids will give strong conjugate bases. A strong base will readily remove a proton from a weak acid.
To state:
How much strong Grignard reagent is and to predict based on the pKa values of alkanes and alkynes whether the reaction shown will occur as written.
b)
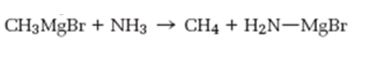
Interpretation:
As a base how much strong Grignard reagent is to be stated. Further Based on the pKa values of alkanes and ammonia to predict whether the reaction shown will occur as written.
Concept introduction:
Stronger acids have low pKa values. A strong acid will yield a weak conjugate base. Similarly weak acids will give strong conjugate bases. A strong base will readily remove a proton from a weak acid.
To state:
How much strong Grignard reagent is and to predict based on the pKa values of alkanes and ammonia whether the reaction shown will occur as written.
Trending nowThis is a popular solution!

Chapter 10 Solutions
Organic Chemistry
- Arrange the following bases in order of increasing strength:Hydrazine (Kb = 1.3x10-6) Pyridine (Kb = 1.6x10-9) Trimethylamine (Kb = 6.4X10-5)arrow_forwardwhich acid of each pair would you expect to be stronger? based on your answer, provide a reaction of the selected stronger acid forming a carboxylate ion #1arrow_forwardwhich acid of each pair would you expect to be stronger? then, provide a reaction of the selected stronger acid forming a carboxylate ion help pls i will upvote #1arrow_forward
- Which has a lower pKa value, the conjugate acid of 3-bromoquinuclidine or the conjugate acid of 3 -chloroquinuclidine?arrow_forwardif the pKa value for p-nitrophenol is 7.08, what fraction of it exists in its ionized form at (a) pH 7, and (b) pH 9?arrow_forward(a) Arrange the following compounds in an increasing order of their indicated property :(i) Benzoic acid, 4-Nitrobenzoic acid, 3,4-Dinitrobenzoic acid, 4-Methoxybenzoic acid (acid strength)(ii) CH3CH2CH (Br) COOH, CH3CH (Br) CH2COOH,(CH3)2CHCOOH, CH3CH2CH2COOH (acid strength)(b) How would you bring about the following conversions :(i) Propanone to Propene (ii) Benzoic acid to Benzaldehyde(iii) Bromobenzene to 1-phenylethanolarrow_forward
- Which of the following reactions will not produce a carboxylic acid? Benzaldehyde + hot, conc'd KMnO4 1) Ph-N2+ + KI 2) BrMgCH=CH2 in ether, followed by H3O+ sec-butylBenzene + hot, conc'd KMnO4 1) Benzene, HNO3 + H2SO4; 2) Fe, HCl; 3) NaNO2; 4) CuCN; 5) dilute acid and heat 1) Benzene and acetyl chloride & AlCl3; 2) HCl & Ni Thank you!arrow_forwardWhich base would you expect to generate the stronger conjugate acid? A.) Hydrazine Kb = 1.7 x 10-4 B.) Analine Kb = 4.3 x 10-10 C.) Triethylamine Kb = 1.0 x 10-3 D.) Ammonia Kb = 1.8 x 10-5arrow_forwardFrom 10 to 14. can i get an explanation on how to choose the appropriate products for these reactions? thank youarrow_forward
- Show a detailed arrow pushing mechanism for the reaction of the Aryl Diazonium Salt with an electron rich aromatic ring to form Orange II(azo dyes) synthesized?arrow_forwardBase from the illustration: 1. Which compound/s will test positive in Baeyer’s test? 2. Which compound/s will produce an orange precipitate upon reaction with 2,4-DNPH? 3. Which compound/s will test positive in the Iodoform test? 4. Which compound/s will produce a brick-red precipitate upon reaction with Fehling’s reagent (CuSO4, tartrate, NaOH)? 5.arrow_forwardInstead of adding to the 4a position and protonating N-5, the thiolate ion could have added to the 10a position and protonated N-1 Why is addition to the 4a position favored?arrow_forward
