
Organic Chemistry
9th Edition
ISBN: 9781305080485
Author: John E. McMurry
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 10.SE, Problem 50AP
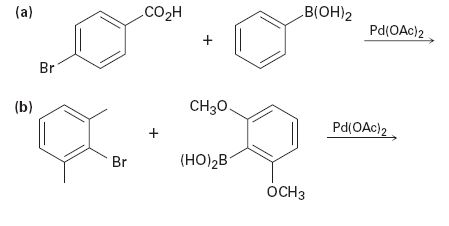
Predict the product and provide the entire catalytic cycle for the Suzuki–Miyaura reactions below.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Conformation control of reaction mechansims.
Consider the reaction below:
Provide all possible elimination products from this reaction and identify the major elimination product. Provide mechanisms (with curved arrows) that clearly explain the reaction outcome.
predict the main product for the addition of 1 equivalent of HX to the following compounds and write the mechanism of the reaction
Give the product/s for the following reaction and indicate what mechanism is involved in the formation of such product/s as SN1, SN2, E1, E2.
CH3CH=CHCl + NaNH2
Chapter 10 Solutions
Organic Chemistry
Ch. 10.1 - Prob. 1PCh. 10.1 - Draw structures corresponding to the following...Ch. 10.2 - Prob. 3PCh. 10.2 - Taking the relative reactivities of 1°, 2°, and...Ch. 10.4 - Prob. 5PCh. 10.4 - The major product of the reaction of...Ch. 10.4 - Prob. 7PCh. 10.5 - Prob. 8PCh. 10.6 - Prob. 9PCh. 10.6 - How might you replace a halogen substituent by a...
Ch. 10.7 - How would you carry out the following...Ch. 10.8 - Rank both sets of compounds in order of increasing...Ch. 10.8 - Tell whether each of the following reactions is an...Ch. 10.SE - Prob. 14VCCh. 10.SE - Prob. 15VCCh. 10.SE - Prob. 16VCCh. 10.SE - Draw the electron-pushing mechanism for each...Ch. 10.SE - Draw the electron-pushing mechanism for the...Ch. 10.SE - The formation of Br2 from NBS first involves the...Ch. 10.SE - In light of the fact that tertiary alkyl halides...Ch. 10.SE - Alkyl halides can be reduced to alkanes by a...Ch. 10.SE - Name the following alkyl halides:Ch. 10.SE - Prob. 23APCh. 10.SE - Draw and name all of the monochlorination products...Ch. 10.SE - How would you prepare the following compounds,...Ch. 10.SE - Prob. 26APCh. 10.SE - A chemist requires a large amount of...Ch. 10.SE - What product(s) would you expect from the reaction...Ch. 10.SE - What product(s) would you expect from the reaction...Ch. 10.SE - What product would you expect from the reaction of...Ch. 10.SE - Rank the compounds in each of the following series...Ch. 10.SE - Which of the following compounds have the same...Ch. 10.SE - Tell whether each of the following reactions is an...Ch. 10.SE - Prob. 34APCh. 10.SE - Alkylbenzenes such as toluene (methylbenzene)...Ch. 10.SE - Prob. 36APCh. 10.SE - Prob. 37APCh. 10.SE - Prob. 38APCh. 10.SE - Prob. 39APCh. 10.SE - Prob. 40APCh. 10.SE - The syntheses shown here are unlikely to occur as...Ch. 10.SE - Why do you suppose its not possible to prepare a...Ch. 10.SE - Prob. 43APCh. 10.SE - Identify the reagents a–c in the following...Ch. 10.SE - Prob. 45APCh. 10.SE - Prob. 46APCh. 10.SE - Prob. 47APCh. 10.SE - The relative rate of radical bromination is...Ch. 10.SE - Prob. 49APCh. 10.SE - Predict the product and provide the entire...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Following is a retrosynthetic scheme for the synthesis of the tricyclic diene on the left. Show how to accomplish this synthesis from 2-bromopropane, cyclopentadiene, and 2-cyclohexenone.arrow_forwardPredict dehydrohalogenation products that results when these alkyl halides are heated in alcoholic KOH. With more than 1 product, predicting the major and minor productsarrow_forwarddraw the expected transition state for the reaction of trans,trans -1,4 diphenyl butadiene with maleic anhydride using the endo rule and predict the stereochemistry of the major qnd minor products from the reaction. help me to draw this..arrow_forward
- 1. Write the complete thermal reaction mechanism to get the compound below. Briefly explain every step. Include starting material, reagents used if there are any and indicate conditions. 2. Write the complete thermal reaction of the compound below with buta-1,3-diene. Indicate reagents and conditions. Provide short explanation every step.arrow_forward3 Give mechanisms for the acid-catalyzed and base-promoted alpha-halogenation ofketones. Explain why multiple halogenations are common with basic catalysis andgive a mechanism for the haloform reactionarrow_forwardConsider the monosubstituted benzene reagent X. Nitration of X yields product Y. Product Y is analysed by 1H and 13C NMR spectroscopy (see Figures 1 to 4). Determine the structure of X and Y on the basis of the spectral data provided.arrow_forward
- Hydration of 1,3-dimethylcyclopentene gives what major and what minor products?arrow_forwardThe aryl diene undergoes sequential Heck reactions to give a product with the molecular formula C15H18. Propose a structural formula for this product.arrow_forwardGive the product/s for the following reaction and indicate what mechanism is involved in the formation of such product/s as SN1, SN2, E1, E2. CH3CHBrCH3 + NaOCH3 in methanolarrow_forward
- 1. Write the complete thermal reaction to get the product below. Include reagents and conditions. Write brief explanation about the reaction mechanism. 2. Write the thermal reaction of the compound below with buta-1,3-diene. Write reagents and conditions. Write brief expanation about the reaction mechanism.arrow_forwardPredict the major product of E1 elimination of the following compounds and write a detailed mechanism for its formationarrow_forwardWhen Br2 is added to buta-1,3-diene at -15 °C, the product mixture contains 60% ofproduct A and 40% of product B. When the same reaction takes place at 60 °C, theproduct ratio is 10% A and 90% B.If you had a solution of pure A, and its temperature were raised to 60 °C, what wouldyou expect to happen? Propose a mechanism to support your prediction.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
IR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=_TmevMf-Zgs;License: Standard YouTube License, CC-BY