
Concept explainers
Interpretation: In the given drawings, the one that best describes the effect of pressure on the solubility of a gas in a liquid is to be identified.
Concept introduction: The Henry law states that the solubility of a gas in a liquid is directly proportional to the pressure of that gas above the surface of the solution.
To determine: The drawing that best describes the effect of pressure on the solubility of a gas in a liquid.
Answer to Problem 11.1VP
Answer
The box (b) states that more gas molecules are soluble at higher pressure and it best explain the effect of pressure on the solubility of a gas in a liquid.
Explanation of Solution
Explanation
The given drawing is,
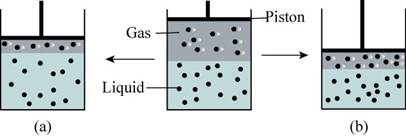
Figure 1
According to Henry’s Law, the solubility of a gas in a liquid is directly proportional to the pressure of that gas above the surface of the solution. When the pressure is increased, the gas molecules are forced into the solution. So, the number of gas molecules decreased as the number of gas molecules dissolved in solution increases.
In box (b), the pressure is increases as the piston is subjected downwards and the gas molecules are dissolved in the solution. Whereas in box (a), the pressure exerted by the piston is small. So lesser is the pressure, lesser will be the solubility of gas molecules in the liquid. Therefore, box (b) best explain the effect of pressure on the solubility of a gas in a liquid.
Conclusion
The box (b) states that more gas molecules are soluble at higher pressure and it best explain the effect of pressure on the solubility of a gas in a liquid.
Want to see more full solutions like this?
Chapter 11 Solutions
Chemistry
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





