
Concept explainers
Interpretation:
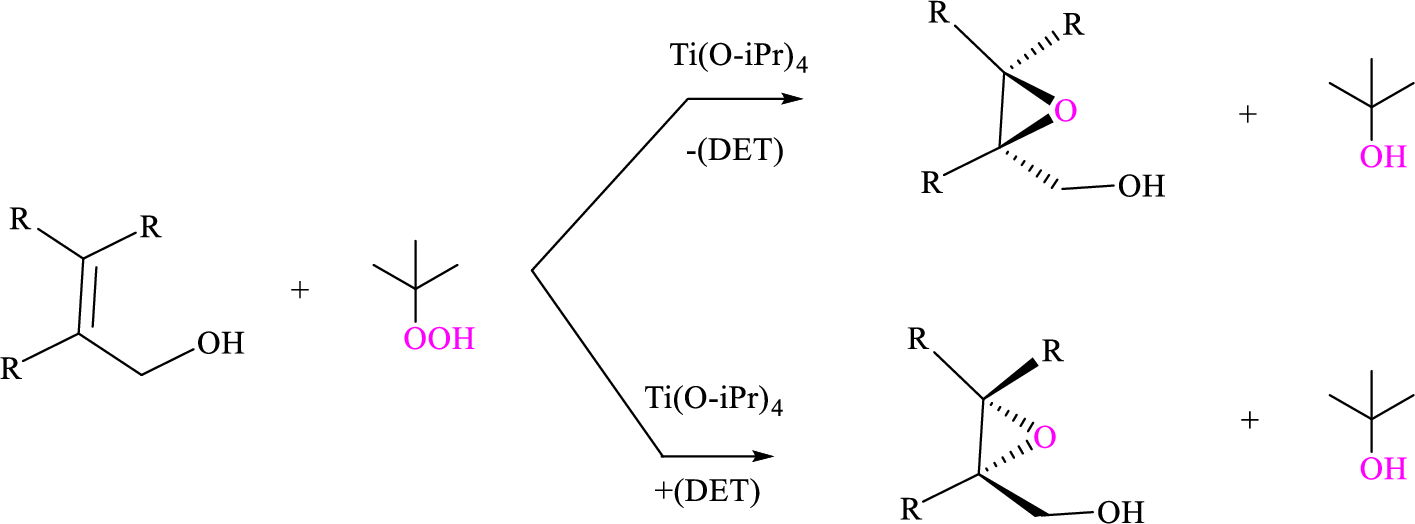
Using Sharpless epoxidation, the allylic alcohol that can be used to prepare the given enantiomer has to be shown.
Concept Introduction:
Sharpless asymmetric epoxidation: Titanium-catalyzed asymmetric epoxidation of primary allylic alcohols is proposed by the Barry Sharpless known as Sharpless epoxidation. The reagent consists of tert-butyl hydroperoxide, titanium tetraisoproxide
Sharpless epoxidation is stereospecific based on the diethyl tartrate
Trending nowThis is a popular solution!

Chapter 11 Solutions
Student Study Guide and Solutions Manual for Brown/Iverson/Anslyn/Foote's Organic Chemistry, 8th Edition
- Nonconjugated , -unsaturated ketones, such as 3-cyclohexenone, are in an acid-catalyzed equilibrium with their conjugated , -unsaturated isomers. Propose a mechanism for this isomerization.arrow_forwardFollowing is the structural formula of Surfynol, a defoaming surfactant. Describe the synthesis of this compound from acetylene and a ketone. How many stereoisomers are possible for Surfynol?arrow_forwardThe first widely used herbicide for the control of weeds was 2,4-dichlorophenoxyacetic acid (2,4-D). Show how this compound might be synthesized from phenol and chloro-acetic acid by way of the given chlorinated phenol intermediate.arrow_forward
- What product is obtained when the following compound undergoes two successive elimination reactions?arrow_forwardWhen trans-2-chloro-1-cyclohexanol is treated with a base, cyclohexene oxide is the product. However, when cis-2-chloro-1-cyclohexanol is treated with a base, the product is cyclohexanone –arrow_forwardThe (R)-isomer of a-terpineol is a component of perfumes and flavorings and has a lilac-like floral odor. The (S)-isomer has a pine-like odor. Propose two methods of producing a-terpineol using Grignard reactions. [Ignore stereochemistry in your synthesises.]arrow_forward
- When cis-2-decalone is dissolved in ether containing a trace of HCI, an equilibrium is established with trans-2-decalone. The latter ketone predominates in the equilibrium mixture. H H HCI cis-2-Decalone trans-2-Decalone Propose a mechanism for this isomerization and account for the fact that the trans iso- mer predominates at equilibrium.arrow_forwardThe following questions concern ethyl (2-oxocyclohexane)carboxylate.(a) Write a chemical equation showing how you could prepare ethyl (2-oxocyclohexane)-carboxylate by a Dieckmann cyclization.(b) Write a chemical equation showing how you could prepare ethyl (2-oxocyclohexane)-carboxylate by acylation of a ketone.(c) Write structural formulas for the two most stable enol forms of ethyl (2-oxocyclohexane)carboxylate.(d) Write the three most stable resonance contributors to the most stable enolate derived from ethyl (2-oxocyclohexane)carboxylate.(e) Show how you could use ethyl (2-oxocyclohexane)carboxylate to prepare 2-methylcyclohexanone.(f) Give the structure of the product formed on treatment of ethyl (2-oxocyclohexane)-carboxylate with acrolein (H2C=CHCH=O) in ethanol in the presence of sodium ethoxidearrow_forwardThe following reaction was performed as part of a research program sponsored by the National Institutes of Health to develop therapeutic agents for the treatment of cocaine addiction. Using what you have seen about the reactions of halogens with alkenes, propose a mechanism for this process.arrow_forward
- Identify products A and B from the given 1H NMR data. Treatment of acetone [(CH3)2C=O] with dilute aqueous base forms B. Compound B exhibits four singlets in its 1H NMR spectrum at 1.3 (6 H), 2.2 (3 H), 2.5 (2 H), and 3.8 (1H) ppm. What is the structure of B?arrow_forwardChoose the correct product and Furnish a step-by-step electron-flow diagram that elucidates the transformation of the product from the enol generated in the second step (that is, illustrate the mechanism by which the enol is converted into a ketone). Explain in detailsarrow_forwardWhat is the role of phosphoric acid in the synthesis of cyclohexene? it is an antioxidant that prevents free radical side reactions it is a safe, non-toxic solvent it lowers the boiling point of the reaction mixture (a colligative property of adding phosphoric acid to water) it protonates the hydroxyl of cyclohexanol to make it a better leaving grouparrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

