
Concept explainers
What reagents are needed to convert
a. 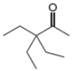 b.
b. 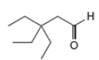 c
c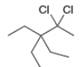 d.
d. 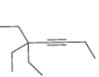
(a)
Interpretation: The reagents that are needed to convert
Concept introduction: A terminal alkyne reacts with
Answer to Problem 11.34P
The reagents that are needed to convert
Explanation of Solution
The reagents that are needed to convert
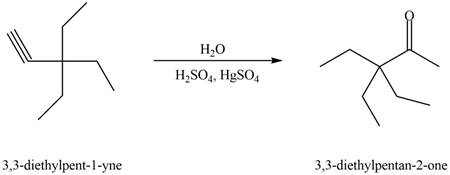
Figure 1
The terminal alkyne,
The reagents that are needed to convert
(b)
Interpretation: The reagents that are needed to convert
Concept introduction: A stepwise procedure of transforming an alkyne into a carbonyl group is known hydroboration-oxidation reaction. In a hydroboration-oxidation reaction, a terminal alkyne reacts with
Answer to Problem 11.34P
The reagents that are needed to convert
Explanation of Solution
The reagents that are needed to convert
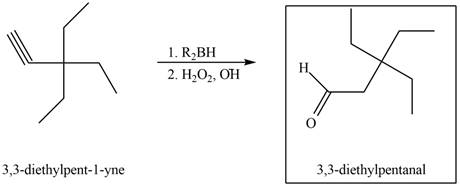
Figure 2
The terminal alkyne,
The reagents that are needed to convert
(c)
Interpretation: The reagents that are needed to convert
Concept introduction: The ellectrophillic reaction which involves the reaction of an alkene or alkyne with
Answer to Problem 11.34P
The reagent that is needed to convert
Explanation of Solution
The reagents that are needed to convert
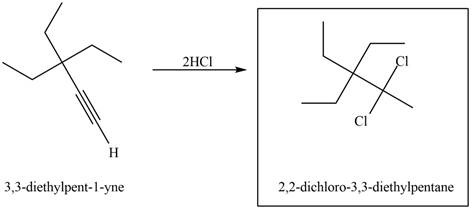
Figure 3
The terminal alkyne,
The reagent that is needed to convert
(d)
Interpretation: The reagents that are needed to convert
Concept introduction: The replacement or substitution of one functional group with another different functional group in any chemical reaction is termed as substitution reaction. The nucleophilic reaction that consists of bimolecular as well as bond-making and bond-breaking steps is termed as
Answer to Problem 11.34P
The reagents that are needed to convert
Explanation of Solution
The reagents that are needed to convert
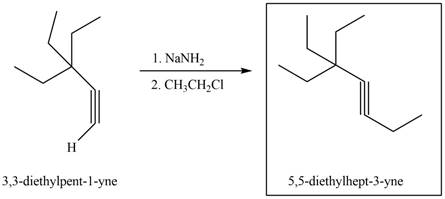
Figure 4
The terminal alkyne,
The reagents that are needed to convert
Want to see more full solutions like this?
Chapter 11 Solutions
ORGANIC CHEMISTRY(LL)W/ACCESS>CUSTOM<
Additional Science Textbook Solutions
Introduction to Chemistry
Chemistry: Atoms First
Introductory Chemistry (6th Edition)
Chemistry: A Molecular Approach
General, Organic, and Biological Chemistry (3rd Edition)
- Draw the product formed when (CH3)2CHOH is treated with each reagent. a. SOCl2, pyridine b. TsCl, pyridine c. H2SO4 d. HBr e. PBr3, then NaCN f. POCl3, pyridinearrow_forwardDraw the products formed when A is treated with each reagent: (a) H2 + Pd-C; (b) mCPBA; (c) PCC; (d) CrO3, H3SO4, H2O; (e) Sharpless reagent with (+)-DET.arrow_forwardDevise a synthesis of each substituted cyclopropane. Use acetylene (HC=CH) as a starting material in part (a) and cyclohexanone as a starting material in part (b). You may use any other organic compounds and any needed reagents.arrow_forward
- (a) Give the IUPAC name for A and B. (b) Draw the product formed when A or B is treated with each reagent: [1] NaBH4, CH3OH; [2] CH3MgBr, then H2O; [3] Ph3P = CHOCH3; [4] CH3CH2CH2NH2, mild acid; [5] HOCH2CH2CH2OH, H+.arrow_forward(a) Give an acceptable name for each compound, (b) Draw the organic products formed when A or B is treated with each reagent: [1] H3O+; [2] −OH, H2O; [3] CH3CH2CH2MgBr (excess), then H2O; [4] LiAlH4, then H2O.arrow_forwardAcyclovir is an effective antiviral agent used to treat the herpes simplexvirus. (a) Draw the enol form of acyclovir, and explain why it is aromatic.(b) Why is acyclovir typically drawn in its keto form, despite the fact thatits enol is aromatic?arrow_forward
- Draw the products formed when A or B is treated with each reagent. In some cases, no reaction occurs. [1] C6H5Li (excess); [2] H2Oarrow_forward(a) Give an acceptable name for compound A. (b) Draw the organic products formed when A is treated with each reagent: [1] H3O+; [2] −OH, H2O; [3] CH3CH2CH2MgBr (excess), then H2O; [4] LiAlH4, then H2O.arrow_forwarda) Draw two different enol tautomers of 2-methylcyclohexanone. (b) Draw two constitutional isomers that are not tautomers, but contain a C = C and an OH group.arrow_forward
- Dimethyl cyclopropanes can be prepared by the reaction of an α,β-unsaturated carbonyl compound X with two equivalents of a Wittig reagent Y. Draw a stepwise mechanism for this reaction.arrow_forwardDimethyl cyclopropanes can be prepared by the reaction of an α, β- unsaturated carbonyl compound X with two equivalents of a Wittig reagent Y. Draw a stepwise mechanism for this reaction.arrow_forward
