
Concept explainers
What
a. 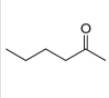 b
b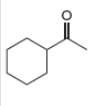 . c.
. c. 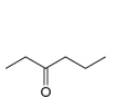 d.
d. 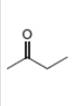
(a)
Interpretation: The alkynes that forms the given ketone as the only product after hydration with
Concept introduction: A terminal alkyne reacts with
Answer to Problem 11.36P
The alkynes that forms the given ketone as the only product after hydration with
Explanation of Solution
The alkynes that forms the given ketone as the only product after hydration with
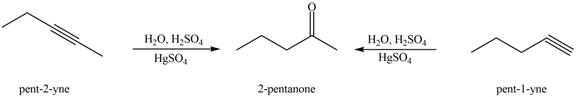
Figure 1
The terminal alkynes,
The alkynes that forms the given ketone as the only product after hydration with
(b)
Interpretation: The alkynes that forms the given ketone as the only product after hydration with
Concept introduction: A terminal alkyne reacts with
Answer to Problem 11.36P
The alkyne that forms the given ketone as the only product after hydration with
Explanation of Solution
The alkyne that forms the given ketone as the only product after hydration with
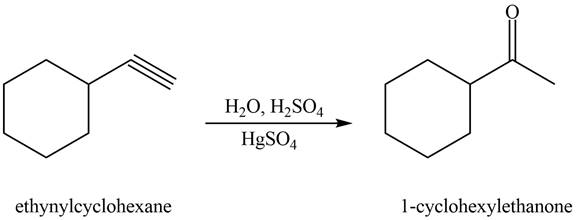
Figure 2
The terminal alkyne, ethynylcyclohexane reacts with the reagents
The alkyne that forms the given ketone as the only product after hydration with
(c)
Interpretation: The alkynes that forms the given ketone as the only product after hydration with
Concept introduction: A terminal alkyne reacts with
Answer to Problem 11.36P
The alkynes that forms the given ketone as the only product after hydration with
Explanation of Solution
The alkynes that forms the given ketone as the only product after hydration with
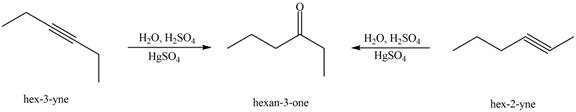
Figure 3
The terminal alkynes,
The alkynes that forms the given ketone as the only product after hydration with
(d)
Interpretation: The alkynes that forms the given ketone as the only product after hydration with
Concept introduction: A terminal alkyne reacts with
Answer to Problem 11.36P
The alkynes that forms the given ketone as the only product after hydration with
Explanation of Solution
The alkynes that forms the given ketone as the only product after hydration with
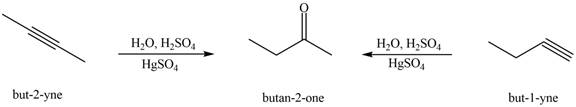
Figure 4
The terminal alkynes,
The alkynes that forms the given ketone as the only product after hydration with
Want to see more full solutions like this?
Chapter 11 Solutions
ORGANIC CHEMISTRY-STUDY GUIDE PACKAGE
Additional Science Textbook Solutions
Chemistry
Chemistry & Chemical Reactivity
Chemistry: The Central Science (13th Edition)
General, Organic, and Biological Chemistry: Structures of Life (5th Edition)
- Draw structural formulas for the alkene that gives each alcohol upon hydroboration-oxidation. (a) (b)arrow_forward1. Ethylene glycol, a major component of antifreeze, becomes especially poisonous when it is oxidized by the liver. Draw two oxidation products that could be derived from ethylene glycol. 2.When 2-methyl-1-butanol is dehydrated in an acid medi- um to an alkene, it yelds mainly 2-methyl-2-butene rather than 2-methyl-1-butene. This indicates that the dehydra- tion to an alkene is at least a two-step reaction. Suggest a mechanism to explain the reaction.arrow_forwardWhat would be the organic product for each reaction?arrow_forward
- give the IUPAC name of the product of 2-bromobutanal that is reacted w/ acidic aqueous KCN?arrow_forwardDraw the overall reaction of 1-butanol with HBr and H2SO4 to form 1-bromobutane.arrow_forwardMaltose is a carbohydrate present in malt, the liquid obtained from barley and other grains. Although maltose has numerous functional groups, its reactions are explained by the same principles we have already encountered.a. Label the acetal and hemiacetal carbons.b. What products are formed when maltose is treated with each of these reagents: [1] H3O+; [2] CH3OH and HCl; [3] excess NaH, then excess CH3I?c. Draw the products formed when the compound formed in Reaction [3] of part (b) is treated with aqueous acid.The reactions in parts (b) and (c) are used to determine structural features of carbohydrates like maltose.arrow_forward
- Draw the products formed when alkene reacts with these reagents: (i) H2 in the presence of Pd catalyst, (ii) HCl.arrow_forwardWhat does alkyl halides gives on heating with dry Ag2O ?arrow_forwardWe have covered several oxidants that use a multi-valent atom (Cr, Cl, S, or I) as their active species, going from a higher oxidation state before the oxidation to a lower oxidation state after oxidizing the alcohol. Draw the structure of the following atoms, before and after the oxidation of an alcohol to a ketone or aldehyde. How many bonds to oxygen does each atom have before and after the oxidation? (a) the I in the DMP reagent (b) the carbinol C in the alcohol that is oxidizedarrow_forward
- Write the structural formula of the organic product for the given reaction between an alkyne and an alkyl halide. The alkyne group is shown and should be entered as "CC" without the triple bond. Enter CC before associated HH atoms (e.g., CH3CH2CH2OCHCHCH3). what is the name of the product. 1.NaNH2 CH3CH2CCH------> product 2. CH3CH2Brarrow_forwardTRUE OR FALSE 9.When n-propyl alcohol undergoes a elimination reaction, the resulting product is an alkene. 10.As the alkyl chain increases in molecular weight among primary alcohol, the solubility in water increases.arrow_forwardFor each of the structures below draw the alcohol that would be predominantly formed from the hydration (an addition reaction) of the double bond. Thank you so much in advance, Chem is aboslutley kicking my a**arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
