
CHEMISTRY F/RADFORD UNIV.W/MASTERI >LL
12th Edition
ISBN: 9781323167359
Author: Timberlake
Publisher: PEARSON C
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 11, Problem 11.44UTC
Convert each of the following line-angle formulas to a condensed structural formula and give its IUPAC name: (11.3)
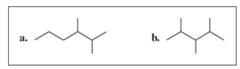
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Draw a condensed structural formula and a line-angle formula for the compound shown below
What is the preferred IUPAC name of the compound in Figure 12. [Numbers are separated by commas and numbers and letters by hyphens. Use lowercase letters and do not use spaces.] *
Your answer
Part A: Draw the condensed structural formula for propyl butyrate
Part B: Draw the condensed structural formula for butyl formate.
Part C: Draw the line-angle formula for ethyl pentanoate:
Chapter 11 Solutions
CHEMISTRY F/RADFORD UNIV.W/MASTERI >LL
Ch. 11.1 - Prob. 11.1QAPCh. 11.1 - Prob. 11.2QAPCh. 11.1 - Identify each of the following properties as more...Ch. 11.1 - Identify each of the following properties as more...Ch. 11.1 - Prob. 11.5QAPCh. 11.1 - Prob. 11.6QAPCh. 11.2 - Give the IUPAC name for each of the following...Ch. 11.2 - Give the IUPAC name for each of the following...Ch. 11.2 - Prob. 11.9QAPCh. 11.2 - Draw the condensed structural formula for alkanes...
Ch. 11.3 - Indicate whether each of the following pairs...Ch. 11.3 - Indicate whether each of the following pairs...Ch. 11.3 - Give the IUPAC name for each of the following a....Ch. 11.3 - Give the TUPAC name for each of the following: a....Ch. 11.3 - Draw the condensed structural formula for each of...Ch. 11.3 - Draw the condensed structural formula for each of...Ch. 11.3 - Draw the line-angle formula for each of the...Ch. 11.3 - Prob. 11.18QAPCh. 11.4 - Heptane, used as a solvent for rubber cement, has...Ch. 11.4 - Nonane has a density of 0.79 g/mL and boils at 151...Ch. 11.4 - Prob. 11.21QAPCh. 11.4 - Prob. 11.22QAPCh. 11.5 - Prob. 11.23QAPCh. 11.5 - Identify the following as alkanes, alkenes,...Ch. 11.5 - Give the IUPAC name for each of the following: a....Ch. 11.5 - Give the IUPAC name for each of the following: a....Ch. 11.5 - Draw the condensed structural formula, or...Ch. 11.5 - Prob. 11.28QAPCh. 11.6 - Prob. 11.29QAPCh. 11.6 - Prob. 11.30QAPCh. 11.6 - Prob. 11.31QAPCh. 11.6 - Prob. 11.32QAPCh. 11.7 - Prob. 11.33QAPCh. 11.7 - Prob. 11.34QAPCh. 11.8 - Prob. 11.35QAPCh. 11.8 - Prob. 11.36QAPCh. 11.8 - Prob. 11.37QAPCh. 11.8 - Prob. 11.38QAPCh. 11 - Prob. 11.39UTCCh. 11 - Prob. 11.40UTCCh. 11 - Prob. 11.41UTCCh. 11 - Prob. 11.42UTCCh. 11 - Prob. 11.43UTCCh. 11 - Convert each of the following line-angle formulas...Ch. 11 - Give the IUPAC name for each of the following:...Ch. 11 - Give the IUPAC name for each of the following:...Ch. 11 - Give the IUPAC name (including cis or trans, if...Ch. 11 - Give the LUPAC name (including cis or trans, if...Ch. 11 - Prob. 11.49AQAPCh. 11 - Prob. 11.50AQAPCh. 11 - Prob. 11.51AQAPCh. 11 - Prob. 11.52AQAPCh. 11 - Draw the condensed structural or line-angle...Ch. 11 - Draw the condensed structural or line-angle...Ch. 11 - Draw the cis and trans isomers for each of the...Ch. 11 - Draw the cis and trans isomers for each of the...Ch. 11 - Prob. 11.57AQAPCh. 11 - Prob. 11.58AQAPCh. 11 - Prob. 11.59AQAPCh. 11 - Prob. 11.60AQAPCh. 11 - Prob. 11.61AQAPCh. 11 - Give the name for the product from the...Ch. 11 - Prob. 11.63AQAPCh. 11 - Prob. 11.64AQAPCh. 11 - Prob. 11.65CQCh. 11 - Prob. 11.66CQCh. 11 - Prob. 11.67CQCh. 11 - Prob. 11.68CQCh. 11 - Prob. 11.69CQCh. 11 - Prob. 11.70CQCh. 11 - Prob. 11.71CQCh. 11 - Margarines are produced from the hydrogenation of...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 5 and 8 only. Write the condensed formulaarrow_forwardDraw the expanded structural formula for the condensed formula CH2=CHCH2CH2CH(CH3)2. Draw all hydrogen atoms.arrow_forwardReview the opening photograph about chocolate (which shows the structure of an active ingredient, theobromine) and then answer the following questions. (a) How do theobromine and caffeine differ structurally? (b) A 5.00-g sample of Hersheys cocoa contains 2.16% theobromine. What is the mass of the compound in the sample?arrow_forward
- Q.N.1 a. Write down the expanded structural formula, condensed structural formula as well as line-angel structural formula for heptane. b.Draw all possible isomers of pentanearrow_forwardThe bond energies of Cl-Cl, C-C and C-H are 59 Kcal/mole, 90 Kcal/mole and 105 Kcal/mole respectively. Considering the reaction of ethane with Cl2, which of the following statements are correct? Question 38 options: A. When heat is the source of energy, Cl-Cl bond is the easiest to break B. When light is the source of energy, Cl-Cl absorbs visible light, but ethane does not C. If heat is the source of energy, the C-H bonds breaks easily compared to other bonds involved in the reaction D. If heat is the source of energy, C-C bonds breaks easily compared to other bonds involved in the reaction Both A & Barrow_forwardIn lab, a student was given a hydrated version of NiCl2 as the unknown. The initial mass of the hydrated salt was 0.782 g. The student heated the crucible for 10 minutes with the cover slightly ajar. After cooling, the dried salt weighed 0.503 g. Based on these results, answer the following questions. What is the chemical formula for this hydrate? What is the IUPAC name for this hydrate?arrow_forward
- For IUPAC name, draw the corresponding structural formula and line-angle formula. Q.)Ethanoic acidarrow_forwardUSE THE CONDENSED STRUCTURAL FORMULA IN WRITING THE ORGANIC COMPOUND AND GIVE THE NECESSARY COEFFICIENT BESIDE EACH COMPOUND IF NEEDED answer number 5 onlyarrow_forward1a. Draw the condensed structural formula for 1-propanamine. 1b. Draw the line-angle formula for 1-propanamine. 2a. Draw the condensed structural formula for 2,3-dimethylheptane 2b. Draw the line-angle formula for 2,3-dimethylheptane 3a. Draw the condensed structural formula for 3-bromo-2-pentanol 3b. Draw the line-angle formula for 3-bromo-2-pentanolarrow_forward
- Circle the letter of the correct answer. 1. Which of the following compounds is saturated? (a) propene (b) octane (c) 3-heptyne (d) 2-butene (e) 1-butene 2. Which formula represents an alkyne? (a) C5H8(b) C5H12(c) C5H16(d) C5H14(e) C5H10 3. What is the total number of covalent bonds in a molecule of C4H10O? (a) 10 (b) 12 (c) 14 (d) 16 (e) 18 4. What is the name of the alkyl group containing three carbon atoms? (a) diphenyl (b) trimethyl (c) propyl (d) methyl (e) butyl 5. What is the IUPAC name of the following molecule? (a) 2-bromo-4-isopropyl-3-methylbutane (b) 2-bromo-3,5-dimethylhexane (c) 3,4-dimethyl-2-bromohexane (d) 2-bromo-3-methyl-4-propylbutane (e) 2-bromo-3,5-dimethylheptane 6. Which of the following compounds is considered to be non-polar? (a) CH3CH2OH (b) CH3CHO (c) CH3COOH (d) CH3CH2NH2(e) CH3CH2CH3 (e) Ethylene glycol is insoluble in water. 7. The molecule CH3COCH3 is classified as: (a) an ether (b) an ester (c) an aldehyde (d) a ketone (e) a carboxylic acid 8.…arrow_forwardIn lab, a student was given a hydrated version of NiCl2 as the unknown. The initial mass of the hydrated salt was 0.821 g. The student heated the crucible for 10 minutes with the cover slightly ajar. After cooling, the dried salt weighed 0.448 g. Based on these results, answer the following questions. What is the chemical formula for this compound? What is the IUPAC name for this hydrate?arrow_forward1a. Draw the condensed structural formula for 1-propanamine. 1b. Draw the line-angle formula for 1-propanamine. 2a. Draw the condensed structural formula for 2,3-dimethylheptane 2b. Draw the line-angle formula for 2,3-dimethylheptane 3a. Draw the condensed structural formula for 3-bromo-2-pentanol 3b. Draw the line-angle formula for 3-bromo-2-pentanol 4a. Draw the condensed structural formula for 3-isopropyl-2-hexene 4b. Draw the line-angle formula for 3-isopropyl-2-hexene 5a. Draw the condensed structural formula for 4-chlorobutanoic acid 5b. Draw the line-angle formula for 4-chlorobutanoic acidarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning
Nomenclature: Crash Course Chemistry #44; Author: CrashCourse;https://www.youtube.com/watch?v=U7wavimfNFE;License: Standard YouTube License, CC-BY