
(a)
Interpretation:
The product of the reaction of
Concept introduction:
Answer to Problem 11.46AP
The product of the reaction of
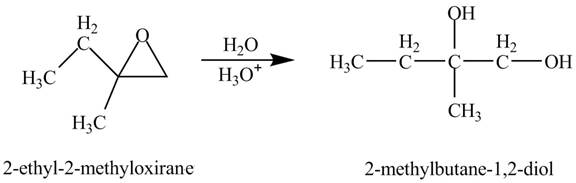
Explanation of Solution
The compound
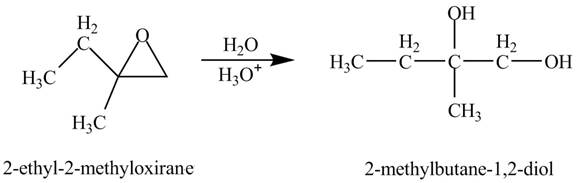
Figure 1
The product of the reaction of
(b)
Interpretation:
The product of the reaction of
Concept introduction:
Epoxides undergo nucleophilic ring opening reactions which are base-catalyzed. If the epoxide is unsymmetrical, then the anionic nucleophile will attack the less-hindered carbon atom of the ring. If the reaction conditions are basic, then the reaction will occur at the less substituted carbon atom.
Answer to Problem 11.46AP
The product of the reaction of
Explanation of Solution
The compound

Figure 2
The product of the reaction of
(c)
Interpretation:
The product of the reaction of
Concept introduction:
Epoxides undergo nucleophilic ring-opening reactions which are base-catalyzed. If the epoxide is unsymmetrical, then the anionic nucleophile will attack the less-hindered carbon atom of the ring. If the reaction conditions are basic, then the reaction will occur at the less substituted carbon atom.
Answer to Problem 11.46AP
The product of the reaction of
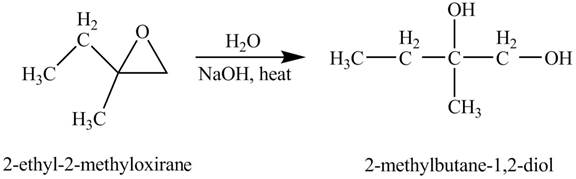
Explanation of Solution
The compound
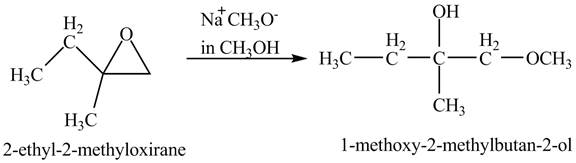
Figure 3
The product of the reaction of
(d)
Interpretation:
The product of the reaction of
Concept introduction:
Epoxides undergo nucleophilic ring-opening reactions which are acid-catalyzed. If the epoxide is unsymmetrical, then the anionic nucleophile will attack the less-hindered carbon atom of the ring. If the reaction conditions are acidic, then the reaction will occur at the more substituted carbon atom.
Answer to Problem 11.46AP
The product of the reaction of
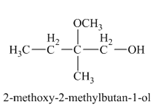
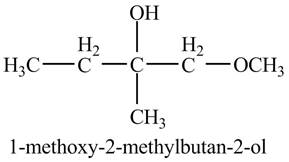
Explanation of Solution
The compound
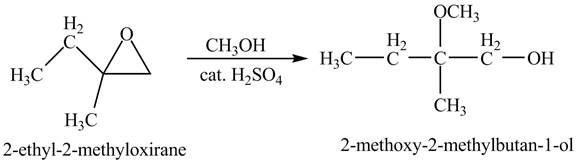
Figure 4
The product of the reaction of
(e)
Interpretation:
The product of the reaction of
Concept introduction:
Epoxides undergo nucleophilic ring opening reactions which are acid-catalyzed. If the epoxide is unsymmetrical, then the anionic nucleophile will attack the less-hindered carbon atom of the ring. If the reaction conditions are acidic, then the reaction will occur at the more substituted carbon atom.
Answer to Problem 11.46AP
The product of the reaction of
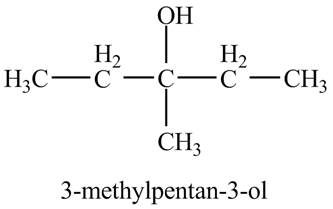
Explanation of Solution
The compound
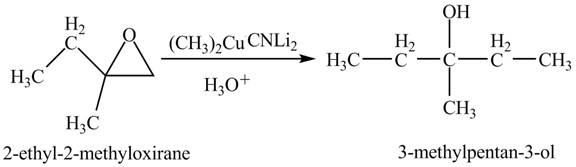
Figure 5
The product of the reaction of
(f)
Interpretation:
The product of the reaction of the product of part (c) with
Concept introduction:
Epoxides undergo nucleophilic ring-opening reactions which are acid-catalyzed. If the epoxide is unsymmetrical, then the anionic nucleophile will attack the less-hindered carbon atom of the ring. If the reaction conditions are acidic, then the reaction will occur at the more substituted carbon atom.
Answer to Problem 11.46AP
The product of the reaction of the product of part (c) with
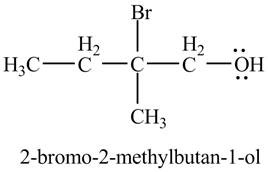
Explanation of Solution
In the presence of
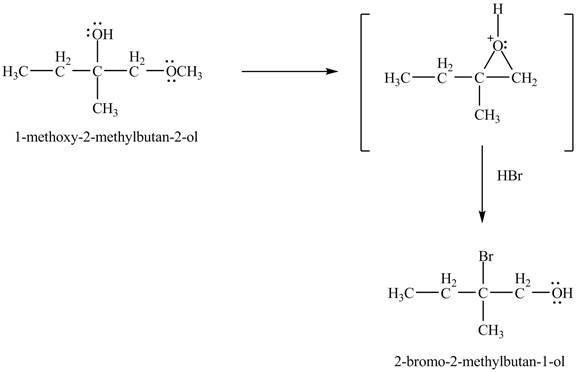
Figure 6
The product of the reaction of the given compound with
(g)
Interpretation:
The product of the reaction of the product of part (d) with
Concept introduction:
Epoxides undergo nucleophilic ring-opening reactions which are acid-catalyzed. If the epoxide is unsymmetrical, then the anionic nucleophile will attack the less-hindered carbon atom of the ring. If the reaction conditions are acidic, then the reaction will occur at the more substituted carbon atom.
Answer to Problem 11.46AP
The product of the reaction of the product of part (d) with
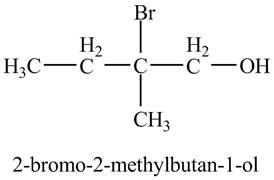
Explanation of Solution
In the presence of
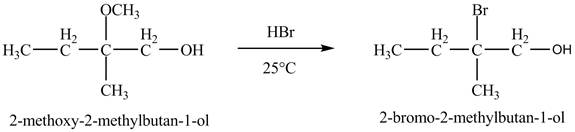
Figure 7
The product of the reaction of the given compound with
(h)
Interpretation:
The product of the reaction of the product of part (c) with
Concept introduction:
The metal hydride reagents are good reducing agents such as
Answer to Problem 11.46AP
The product of the reaction of the product of part (c) with
Explanation of Solution
The base
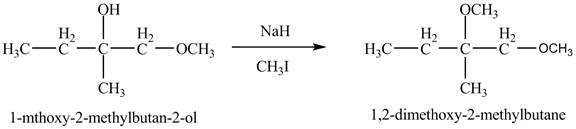
Figure 8
The product of the reaction of the given compound with
(i)
Interpretation:
The product of the reaction of the product of part (d) with
Concept introduction:
The metal hydride reagents are good reducing agents such as
Answer to Problem 11.46AP
The product of the reaction of the product of part (d) with
Explanation of Solution
The base
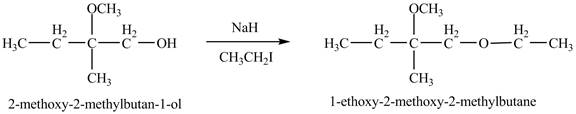
Figure 9
The product of the reaction of the given compound with
(j)
Interpretation:
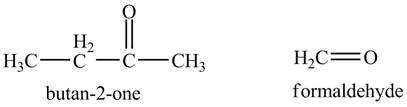
The product of the reaction of the product of part (a) with periodic acid is to be predicted.
Concept introduction:
The periodic acid acts as a strong oxidizing agent. The periodic acid reacts with a vicinal diol to form two
Answer to Problem 11.46AP
The products of the reaction of the product of part (a) with periodic acid are shown below.
Explanation of Solution
The given compound is vicinal diol. It reacts with periodic acid to form two aldehydes. The carbon-carbon bond between the carbon atoms attached to two adjacent hydroxyl groups gets breaks. The corresponding chemical reaction is shown below.
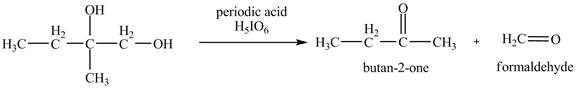
Figure 10
The products of the reaction of the given compound with periodic acid are shown in Figure 10.
(k)
Interpretation:
The product of the reaction of the product of part (f) with
Concept introduction:
Grignard reagents are
Answer to Problem 11.46AP
The product of the reaction of the product of part (f) with
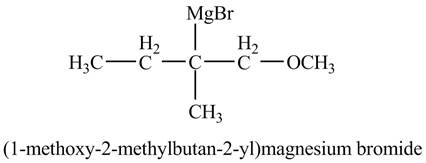
Explanation of Solution
The compound
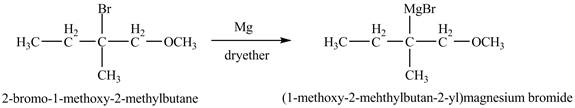
Figure 11
The product of the reaction of the given compound with
(l)
Interpretation:
The product of the reaction of the product of part (k) with ethylene oxide followed by addition of
Concept introduction:
Grignard reagents are organometallic compounds which are prepared using alkyl halides in the presence of magnesium metal in dry ether. These reagents act as strong nucleophiles and bases. Grignard reagents react with carbonyl compounds to form alcohol.
Answer to Problem 11.46AP
The product of the reaction of the product of part (k) with ethylene oxide followed by addition of
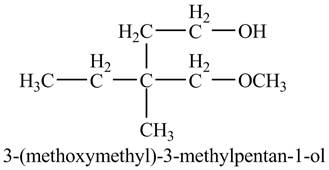
Explanation of Solution
Grignard reagent can act as a nucleophile. In the presence of an acid, it can attack the more substituted carbon atom of the epoxy ring. The Grignard reagent reacts with the ethylene oxide followed by protonolysis to form alcohol. The corresponding chemical reaction is shown below.
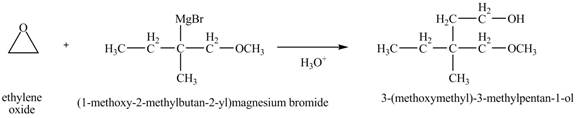
Figure 12
The product of the reaction of the given compound with ethylene oxide followed by addition of
Want to see more full solutions like this?
Chapter 11 Solutions
EBK ORGANIC CHEMISTRY STUDY GUIDE AND S
- 6) Which is the organic product for the following reaction? (a) (b) (c) (d) сон COOH ОН ОН COOH COOH KMnO4 H2Oarrow_forward6) Which is the organic product for the following reaction? (a) (b) (c) (d) LOH OH COOH OH OH COOH COOH KMnO4 H₂O (e) None of the above products will be formedarrow_forward(b) 3-methyl-2-butanol reacts with concentrated sulphuric acid to form 2-methyl-2- butene. Write the mechanism for the reaction.arrow_forward
- (a) How will you convert:(i) Benzene to acetophenone (ii) Propanone to 2-Methylpropan-2-ol(b) Give reasons :(i) Electrophilic substitution in benzoic acid takes place at meta position.(ii) Carboxylic acids are higher boiling liquids than aldehydes, ketones and alcohols of comparable molecular masses.(iii) Propanal is more reactive than propanone in nucleophilic addition reactions.arrow_forwardName the type of reaction; then predict the products. Make sure the reaction is balanced.arrow_forwardIn addition: can u give the names of evey compounds in the question solutionarrow_forward
- provide the structure of the intermediate and product for the following reaction : (c) H CH,OH/H (C)arrow_forwardWrite the products of the following acid-base reactions: (a) CH3OH + H2SO4 ² ? (b) CH3OH + NANH2 2 ? (c) CH3NH3+ Cl- + NaOH ?arrow_forwardDraw the reaction products of the following reactions of the compound 1-pentanol and name the compounds according to IUPAC! (a) B-elimination of H20 (b) SN reaction in the presence of hydrochloric acid (c) Condensation with acetic acid (d) Oxidation to the corresponding carbonyl compound (e) Condensation of two butanol moleculesarrow_forward
- Give an IUPAC and common name for each of the following naturally occurring carboxylic acids: (a) CH3CH(OH)CO2H (lactic acid); (b) HOCH2CH2C(OH)(CH3)CH2CO2H (mevalonic acid).arrow_forwardGive reasons for the following: (i) p-nitrophenol is more acidic than p-methylphenol. (ii) Bond length of C—O bond in phenol is shorter than that in methanol. (iii) (CH3)3C—Br on reaction with sodium methoxide (Na+ _OCH3) gives alkene as the main product and not an ether.arrow_forwardDraw line structures of the following compounds and the product you would obtain from the reduction of each.(a) Isopropyl methyl ketone (b) p-Hydroxybenzaldehyde(c) 2-Methylcyclopentanonearrow_forward
