
Concept explainers
(a)
Interpretation:
The product of the reaction of dibutyl sulfide with
Concept introduction:
Hydrogen perioxide
Sulfur containing organic compounds has many similar chemical properties as oxygen contain compound such as ether and aclcohol.
Answer to Problem 11.45AP
The product of the reaction of dibutyl sulfide with

Explanation of Solution
The dibutyl sulfide undergoes an oxidation reaction with hydrogen peroxide. The dibutyl sulfide reacts with
The corresponding

Figure 1
The product of the reaction of dibutyl sulfide with
(b)
Interpretation:
The product of the reaction of dibutyl sulfide with
Concept introduction:
Hydrogen perioxide
Sulfur containing organic compounds has many similar chemical properties as oxygen contain compound such as ether and aclcohol.
Answer to Problem 11.45AP
The product of the reaction of dibutyl sulfide with
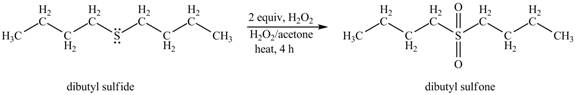
Explanation of Solution
The dibutyl sulfide undergoes an oxidation reaction with hydrogen peroxide. The dibutyl sulfide reacts with
The corresponding chemical reaction is shown below.
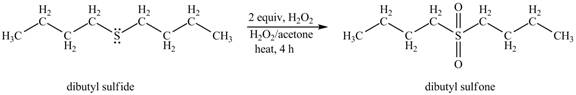
Figure 2
The product of the reaction of dibutyl sulfide with
(c)
Interpretation:
The product of the reaction of
Concept introduction:
The magnesium monoperoxyphthalate
Answer to Problem 11.45AP
The product of the reaction of
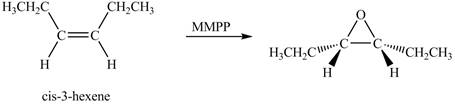
Explanation of Solution
The compound
The corresponding chemical reaction is shown below.
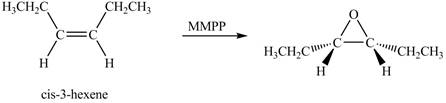
Figure 3
The product of the reaction of
(d)
Interpretation:
The product of the reaction of given compound with
Concept introduction:
Epoxides undergo nucleophilic ring-opening reactions which are acid-catalyzed. If the epoxide is unsymmetrical, then the anionic nucleophile will attack the less-hindered carbon atom of the ring. If the reaction conditions are acidic, then the reaction will occur at the more substituted carbon atom.
Answer to Problem 11.45AP
The product of the reaction of given compound with
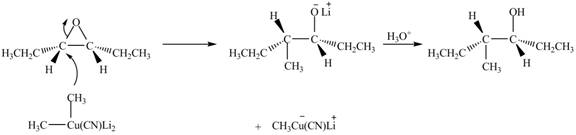
Explanation of Solution
The epoxide undergoes ring-opening reaction in the presence of acid. The dilithium dimethylcyanocuprate molecule generates a nucleophile
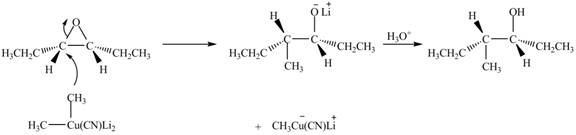
Figure 4
The product of the reaction of given compound with
(e)
Interpretation:
The product of the reaction of given compound with solvent
Concept introduction:
Epoxides undergo nucleophilic ring-opening reactions which are acid-catalyzed. If the epoxide is unsymmetrical, then the anionic nucleophile will attack the less-hindered carbon atom of the ring. If the reaction conditions are acidic, then the reaction will occur at the more substituted carbon atom.
Answer to Problem 11.45AP
The product of the reaction of given compound with solvent
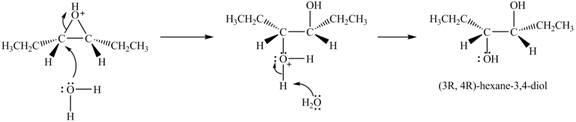
Explanation of Solution
The epoxide undergoes ring opening reaction in the presence of acid. The water molecule acts a nucleophile and attacks on the more substituted carbon atom of the epoxide ring to from

Figure 5
The product of the reaction of given compound with solvent
(f)
Interpretation:
The product of the reaction of given compound with periodic acid is to be predicated.
Concept introduction:
The periodic acid acts as a strong oxidizing agent. The periodic acid reacts with a vicinal diol to form two
Answer to Problem 11.45AP
The product of the reaction of given compound with periodic acid is shown below.
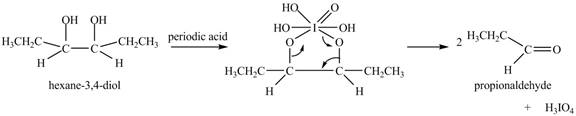
Explanation of Solution
The given compound is vicinal diol. It reacts with periodic acid to form two aldehydes. The carbon-carbon bond between the carbon atoms attached to two adjacent hydroxyl groups gets breaks. The corresponding chemical reaction is shown below.

Figure 6
The product of the reaction of given compound with periodic acid is shown in Figure 6.
(g)
Interpretation:
The product of the reaction of given compound with
Concept introduction:
The metal hydride reagents are good reducing agents such as
Answer to Problem 11.45AP
The product of the reaction of given compound with
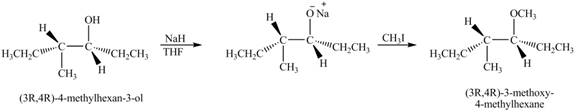
Explanation of Solution
The base
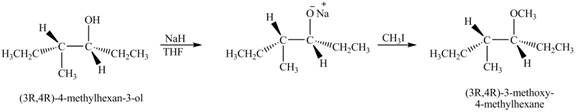
Figure 7
The product of the reaction of given compound with
(h)
Interpretation:
The product of the reaction of
Concept introduction:
Oxymercuration reaction is a type of reaction in which an alkene get converted into an alcohol. The mercuric acetate is used in the reaction as reagent. This reagent attacks the alkene to form a cyclic intermediate compound which further undergoes reduction to form alcohol.
Answer to Problem 11.45AP
The product of the reaction of
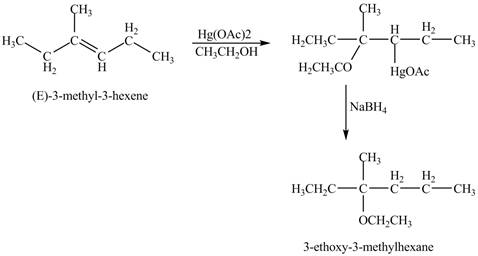
Explanation of Solution
The mercuric acetate
The corresponding chemical reaction is shown below.
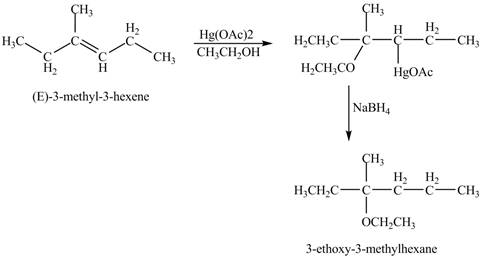
Figure 8
The product of the reaction of
(i)
Interpretation:
The product of the reaction of the given compound with acidic methanol is to be predicated.
Concept introduction:
The replacement or substitution of one functional group with another different functional group in any chemical reaction is termed as a substitution reaction. In a nucleophilic substitution reaction, nucleophile takes the position of leaving the group by attacking the electron-deficient carbon atom.
Answer to Problem 11.45AP
The product of the reaction of the given compound with acidic methanol is shown below.
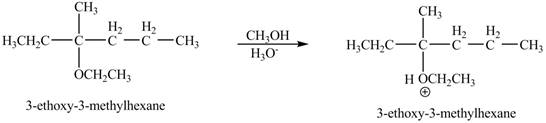
Explanation of Solution
The proton of the acid will protionate the ether. The protonated ether will increase the electrophilic characters of carbon atom attached to the oxygen atom. The methonal can acts as nuclephile, however the nucleophilic charater of methoxy-group and ethoxy group is similar. Therefore, no further reaction will take place.
The corresponding chemical reaction is shown below.
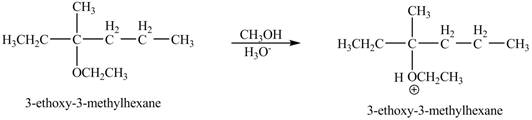
Figure 9
The product of the reaction of the given compound with acidic methanol is shown in Figure 9.
(j)
Interpretation:
The product of the reaction of
Concept introduction:
Grignard reagents are
Answer to Problem 11.45AP
The product of the reaction of
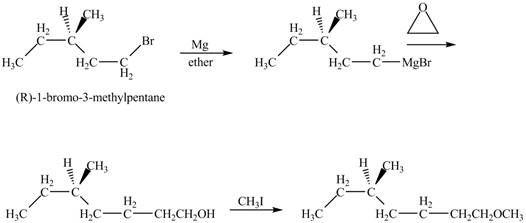
Explanation of Solution
The alkyl halide will react with magnisum metal to from gridnard reagent. These Grignard reagent reacts with epoixde to form alcohol. The alcohol reacts with
The corresponding chemical reaction is shown below.
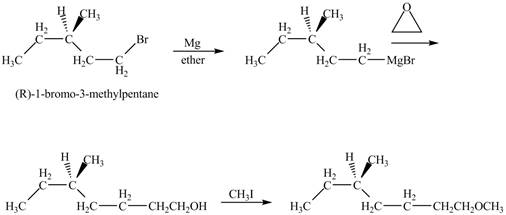
Figure 10
The product of the reaction of
Want to see more full solutions like this?
Chapter 11 Solutions
EBK ORGANIC CHEMISTRY STUDY GUIDE AND S
- Answer ALL parts. (a) Treatment of enone C with tetrabutylammonium fluoride leads to the formation of heterocycle D. 2 steps BU4NF C9H1602 A + B D OSIME,tBu (i) Enone C can be prepared from alkyne A and aldehyde B in two steps via a gold-catalysed reaction. Identify A and B and provide reagents/conditions for a synthesis of C. (ii) Give the structure of D and classify the ring-closing reaction for its formation from C according to Baldwin's rules (iii) Sketch the key molecular orbitals involved in the ring-closing reaction in part (ii) and hence explain whether the reaction is favourable or unfavourable.arrow_forwardAccount for the following :(a) The dipole moment of chlorobenzene is lower than that of cyclohexyl chloride.(b) Alkyl halides, though polar, are immiscible with water.(c) Grignard’s reagents should be prepared under anhydrous conditions.arrow_forwardWrite the structure of the major organic product formed in the reaction of 1-pentene with each of the following: (a) Hydrogen chloride (b) Dilute sulfuric acid (c) Diborane in diglyme, followed by basic hydrogen peroxide (d) Bromine in carbon tetrachloride (e) Bromine in water (f) Peroxyacetic acid (g) Ozone (h) Product of part (g) treated with zinc and water (i) Product of part (g) treated with dimethyl sulfide (CH3)2Sarrow_forward
- Starting exactly with any acid chloride with exactly with 5 carbon atoms, and using appropriate reagents outline the synthesis of the following molecules: (a) 2,6-dimethyl-4-heptanone (b) 4-propyl-4-octanolarrow_forward(a) How will you carry out the following conversions?(i) Acetylene to Acetic acid (ii) Toluene to m-nitrobenzoic acid(iii) Ethanol to Acetone(b) Give reasons :(i) Chloroacetic acid is stronger than acetic acid.(ii) pH of reaction should be carefully controlled while preparing ammonia derivatives of carbonyl compounds.arrow_forwardPredict the principal organic product of each of the following reactions. Specify stereochemistry where appropriate.arrow_forward
- 2. (a) Reaction of an alkene with ozone followed by an oxidative workup gives the product shown. What is the structure of alkene A? A (b) Compounds B and C both have the formula C10H16. Hydrogenation of either compound over Pt gives the same product, cis-1-isopropyl-4-methylcyclohexane. Ozonolysis with reductive workup fragments the two compounds differently, as shown below. What are the structures of C B and C? (CHCl3, chloroform, is the solvent.) B 1) 03, CHCI 3 2) (CH3)2S 1) 03, CHC13 2) (CH3)2S H O 1) 03 2) H₂O2, H₂O H3C- H O CH3 3. (a) Draw the structures of all enols that would spontaneously form this ketone, including stereoisomers. + + I HO₂C H + La CH3CH₂-C-CH(CH3)2 O= (b) Would alkyne hydration be a good preparative method for this compound? If so, give the reaction. If not, explain why.arrow_forward(a) Aniline is oxidized and then resulting product is boiled with Conc. HNO3 and conc. H2S04. (b) Cyclopentylnitrile is reduced with LIAIH4 and the resulting product reacts with benzenesulphonylchloride (c) 2-Chloro-5-methyl-diethylhexandioate is condensed in alkaline medium (d) 2-Methylethylpropanoate is condensed with phenylbenzanoate in an alkaline medium (e) When Ethanoic is heated with NH3 and resulting product reacts with benzoylchloride.arrow_forward1arrow_forward
- (a) Illustrate the following name reactions giving a chemical equation in each case :(i) Clemmensen reaction (ii) Cannizzaro’s reaction(b) Describe how the following conversions can be brought about :(i) Cyclohexanol to cyclohexan-1-one (ii) Ethylbenzene to benzoic acid(iii) Bromobenzene to benzoic acidarrow_forwardGive reasons: (a) n-Butyl bromide has higher boiling point than f-butyl bromide. (b) Racemic mixture is optically inactive. (c) The presence of nitro group (-N02) at o/p positions increases the reactivity of haloarenes towards nucleophilic substitution reactionsarrow_forwardGive to all parts? (a) Give the complete mechanism for the reaction of trans-2-butene with Br2 in excess H2O. (b) Name the products (including stereochemistry). (c) Give the complete mechanism for the reaction of trans-2-butene with Br2 in excess H2O. (d) Name the products (including stereochemistry).arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





