
Concept explainers
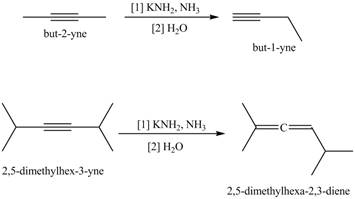
Write a stepwise mechanism for each of the following reactions. Explain why a more stable
Want to see the full answer?
Check out a sample textbook solution
Chapter 11 Solutions
Organic Chemistry
Additional Science Textbook Solutions
Chemistry
Chemistry
Chemistry: Matter and Change
Inorganic Chemistry
Basic Chemistry
Chemistry For Changing Times (14th Edition)
- When toluene is treated with sulfuric and nitric acids under special conditions, three nitro (NO2) groups are substituted for hydrogens at the 2, 4 and 6 positions on the ring (the next section discusses why the 2, 4, and 6 positions are substituted). The product is a highly explosive substance called 2,4,6-trinitrotoluene. This subastance is commonly known by a three letter name. What is it?arrow_forwardWhat synthetic goal is achieved by subjecting an alkene to an oxymercuration-demercuration reaction sequence? anti-Markovnikov addition of H 2O, preventing skeletal rearrangement anti-Markovnikov addition of H2O, promoting skeletal rearrangement Markovnikov addition of H 2O, preventing skeletal rearrangement Markovnikov addition of H 2O, promoting skeletal rearrangement anti-Markovnikov addition of H2O, syn-hydroxylationarrow_forwardOrganic Chemistry Acid catalyzed dehydration reaction of 2-methyl-1-butanol produces 2-methyl-2-butene as the major product. Also acid catalyzed dehydration reaction of 3-methyl-1-butanol give the same product as major product. Explain the reason why both of the reaction produce the same product as the major product.arrow_forward
- complete the conversion using SN1, SN2, E1, or E2. what is a possible reagent? cyclohexane --> cyclodi-1, 3- enearrow_forwardAlkynes generally undergo nucleophilic addition reactions. Ozonolysis of alkynes with O3, Zn and H3O+ yields either a carboxylic acid or carbon dioxide (or both). A. Both statements are true. B. Both statements are false. C. Only the first statement is true. D. Only the second statement is true.arrow_forwardConsider the structure of cyclohexyne, if it undergoes acetylide reaction with NaNH2 and 2 moles of CH3Br, which of the following final product is formed? a. 1,2-methylcyclohexyne HBr b. 1-methylcyclohexyne c. No reactionarrow_forward
- Draw the substitution product formed when cis-1-bromo-4-methylcyclohexane and hydroxide ion undergo an SN2 reaction.arrow_forward1. What are the various ways by which alkenes may be synthesized?2. Give two examples each of Unsymmetrical alkenes and reagents.3. Give two examples of reactions of alkenes that result in Anti-Markonikov’s addition productsarrow_forwardAre terminal alkynes considered weak or strong acids? Explain why...arrow_forward
- 1. Which molecule reacts fastest with ethanol by SN1 mechanism? a. 1-bromopentane b. 3-bromopentane c. 2-bromo-2-methylbutane d. 1-bromo-2,2-dimethylpropanearrow_forwardThe 1,2‑dibromide is synthesized from an alkene starting material. Draw the alkene starting material. Clearly, show stereochemistry of the alkene.arrow_forwardOrder the compounds in each set according to their sn2-displacement reactivity:2-bromo2-methyl butane; 1-bromopentane; 2-bromopentane.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
