
Package: Organic Chemistry With Connect 2-semester Access Card
4th Edition
ISBN: 9781259671838
Author: Janice Gorzynski Smith Dr.
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 11, Problem 11.68P
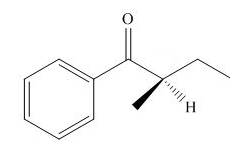
Explain why an optically active solution of
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Explain why a base can remove a proton from the alpha-carbon of N,N-dimethylethanamide but not from the alpha-carbon of either N-methylethanamide or ethanamide.
Explain why a base can remove a proton from the a-carbon of N,N-dimethylethanamide but not from the α-carbon of either N-methylethanamide or ethanamide.
Explain why a proton can be removed from the 'alpha-carbon of N,N-dimethylethanamide but not from the alpha-carbon of either N-methylethanamide or ethanamide.
Chapter 11 Solutions
Package: Organic Chemistry With Connect 2-semester Access Card
Ch. 11 - Draw structures for the three alkynes having...Ch. 11 - Prob. 11.2PCh. 11 - Give the IUPAC name for each compound.Ch. 11 - Give the structures corresponding to each of the...Ch. 11 - Prob. 11.5PCh. 11 - Prob. 11.6PCh. 11 - Which bases can deprotonate acetylene? The pKa...Ch. 11 - Draw the organic products formed when each alkyne...Ch. 11 - Prob. 11.9PCh. 11 - Problem 11.9 Draw the products formed when is...
Ch. 11 - Explain the following result. Although alkenes...Ch. 11 - Problem 11.11 Draw the keto tautomer of each...Ch. 11 - Prob. 11.13PCh. 11 - a Draw two different enol tautomers of...Ch. 11 - Prob. 11.15PCh. 11 - Prob. 11.16PCh. 11 - Prob. 11.17PCh. 11 - Problem. 11.17 Show how , and can be used to...Ch. 11 - Prob. 11.19PCh. 11 - Draw the products of each reaction. a. b.Ch. 11 - Prob. 11.21PCh. 11 - Problem 11.21 Use retrosynthetic analysis to show...Ch. 11 - Prob. 11.23PCh. 11 - Give the IUPAC name for each compound. a. b.Ch. 11 - Prob. 11.25PCh. 11 - 11.25 Answer the following questions about...Ch. 11 - Prob. 11.27PCh. 11 - Give the IUPAC name for each alkyne.Ch. 11 - Prob. 11.29PCh. 11 - Which of the following pairs of compounds...Ch. 11 - Prob. 11.31PCh. 11 - 11.30 How is each compound related to A? Choose...Ch. 11 - Prob. 11.33PCh. 11 - Prob. 11.34PCh. 11 - Prob. 11.35PCh. 11 - 11.33 Draw the products formed when is treated...Ch. 11 - Draw the products formed when 3-hexyne is treated...Ch. 11 - Prob. 11.38PCh. 11 - Prob. 11.39PCh. 11 - What alkynes give each of the following ketones as...Ch. 11 - Prob. 11.41PCh. 11 - 11.37 What alkyne gives each compound as the only...Ch. 11 - Prob. 11.43PCh. 11 - Draw the structure of compounds A-E in the...Ch. 11 - Prob. 11.45PCh. 11 - Prob. 11.46PCh. 11 - 11.42 What reactions are needed to convert alcohol...Ch. 11 - Prob. 11.48PCh. 11 - Prob. 11.49PCh. 11 - 11.45 Explain the following statement. Although ...Ch. 11 - Draw a stepwise mechanism for the following...Ch. 11 - Prob. 11.52PCh. 11 - Prob. 11.53PCh. 11 - Prob. 11.54PCh. 11 - Prob. 11.55PCh. 11 - Synthesize each compound from acetylene. You may...Ch. 11 - Prob. 11.57PCh. 11 - Prob. 11.58PCh. 11 - 11.55 Devise a synthesis of the ketone, , from ...Ch. 11 - 11.56 Devise a synthesis of each compound using ...Ch. 11 - Prob. 11.61PCh. 11 - Prob. 11.62PCh. 11 - 11.60 Draw a stepwise mechanism for the following...Ch. 11 - Draw a stepwise mechanism for the following...Ch. 11 - Prob. 11.65PCh. 11 - Write a stepwise mechanism for each of the...Ch. 11 - Prob. 11.67PCh. 11 - 11.65 Explain why an optically active solution of ...
Additional Science Textbook Solutions
Find more solutions based on key concepts
Q1. What is the empirical formula of a compound with the molecular formula
Chemistry: A Molecular Approach (4th Edition)
For each of the following 2-dimensional shapes, determine the highest order rotation axis of symmetry.
Inorganic Chemistry
Practice Problem 1.22 Which of the following alkenes can exist as cis-trans isomers? Write their structures. Bu...
Organic Chemistry
Practice Problem ATTEMPT
Write the rate expressions for each of the following reactions:
(a)
(b)
(c)
Chemistry
The method to determine the volume of a powered solid, liquid and a rock needs to be determined. Concept introd...
Living by Chemistry
Practice Exercise 1
Which of the following factors determines the size of an atom? a. the volume of the nucleus...
Chemistry: The Central Science (14th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- A racemic mixture of 2-methyl-1-phenyl-1-butanone is formed when (R)-2-methyl-1-phenyl-1-butanone is dissolved in an acidic or basic aqueoussolution. Give an example of another ketone that undergoes acid- or base-catalyzed racemization.arrow_forwardIs 4-Methylphenyl 4-bromobenzoate an alpha- beta conjugated ester?arrow_forwardWhat is a reasonable synthetic pathway on how benzoyl chloride can be synthesized to form N-(2-phenylethyl) aniline?arrow_forward
- what is the structure of o-methylphenoxide anion and what is its pKa?arrow_forwarda. Compound X is benzene, Y is acetic anhydride acid. Complete the following scheme and determine Z! b. Determine which reagents except acetic acid anhydrides can replace Y!arrow_forwardWhat is the major product of bromination of p-chlorobenzoic acid reactions?arrow_forward
- Write reactions of aniline with the following reagents: a. CH3COCl b. Br2 c. CH3Iarrow_forwardp-Nitrobenzaldehyde is more reactive toward nucleophilic additions than p-methoxybenzaldehyde. Explain.arrow_forwardExplain why the ether obtained by treating an optically active alcohol with PBr3 in pyridine followed by sodium methoxide has the same configuration as the alcohol, whereas the ether obtained by treating the alcohol with tosyl chloride followed by sodium methoxide has a configuration opposite that of the alcohol.arrow_forward
- please give the structure of the following molecules: a) t-butyl acetate enolate anion b) N-methylimidazole c) p-toluenesulfonate anion d) phenoxide anionarrow_forwardRank the following compounds in order of increasing basicity: I. p-nitroaniline III. N-methylaniline II. p-aminobenzaldehyde IV. p-methylanilinearrow_forwardWhat is the product resulted due to the reaction of 2-hydroxy-3-methoxybenzaldehyde with ethyl bromoacetate in a basic solution indicating all the reagents and intermediates occurred during the reaction?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
IR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=_TmevMf-Zgs;License: Standard YouTube License, CC-BY