
Concept explainers
(a)
Interpretation:
The structure of the alkene that could have been used to produce deuterated bromoalkene when treated with deuterium bromide (
Concept introduction:
The electrophilic addition of a Bronsted acid to the carbon-carbon double bond in
Answer to Problem 11.8P
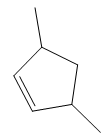
The structure of the corresponding alkene that has been used to produce the given deuterated bromoalkane is
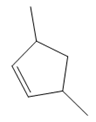
Explanation of Solution
The structure of the given bromoalkane is
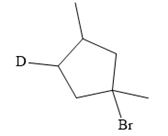
In the structure above, the carbon atom to which the bromine atom is attached must be the carbon bearing the positive charge. Thus, a stable tertiary carbocation must have been formed during the mechanism. Deuterium is an isotope of hydrogen having similar chemical properties. When an alkene is treated with deuterium bromide, the first step of addition is that the deuterium gets attached to the doubly bonded carbon atom which has got a higher number of hydrogens attached (or least substituted). Thus, the carbon atom to which deuterium is attached must be having a double bond in the original alkene. The resulting carbocation that would form will be secondary and can rearrange to a more stable, tertiary carbocation via 1, 2-hydride shift. Thus, a less stable carbocation undergoes carbocation rearrangement reaction to form the most stable carbocation. In the next step, the bromine ion attacks this tertiary carbocation to form the given product. Thus, the structure of the corresponding alkene that could have been used to produce the given deuterated bromoalkane must be
The complete mechanism is shown below:
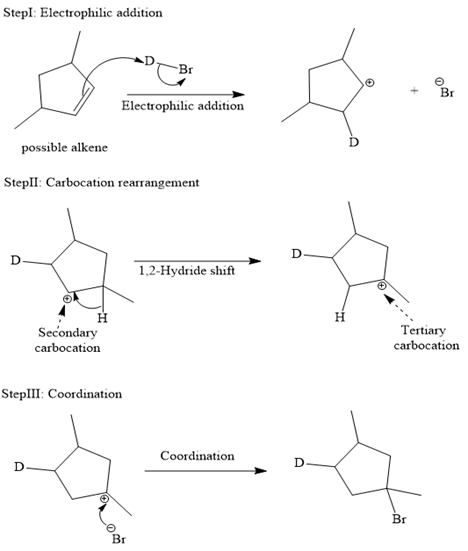
In an electrophilic addition reaction, the carbocation rearrangement reaction takes place to form a rearranged product.
(b)
Interpretation:
The structure of the alkene that could have been used to produce deuterated bromoalkene when treated with deuterium bromide (
Concept introduction:
The electrophilic addition of a Bronsted acid to the carbon-carbon double bond in alkenes is susceptible to carbocation rearrangements due to the stability of the carbocation. The carbocation rearrangement occurs via either 1, 2 hydride shift or 1, 2 methyl shift depending on the stability of the carbocation formed. The stability order for carbocation is benzylic > tertiary > secondary > primary > methyl etc. The normal electrophilic addition gives 1, 2-addition product, but due to the rearrangement reaction, the rearranged product may be formed instead of 1, 2-addition product. In the reaction of an alkene and hydrogen halide or deuterium halide, the carbon to which the deuterium is attached is the carbon bearing the positive charge. Further, the carbon atom to which the halide atom is attached must be the carbon bearing the positive charge, that is, carbocation intermediate. Rearrangements of the carbocation intermediate may be observed if a more stable carbocation is possible.
Answer to Problem 11.8P
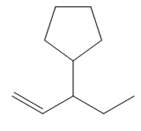
The structure of the corresponding alkene that has been used to produce the given deuterated bromoalkane is
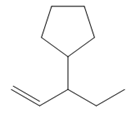
Explanation of Solution
The structure of the given bromoalkane is
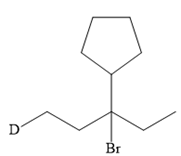
In the structure above, the carbon atom to which the bromine atom is attached must be the carbon bearing the positive charge. Thus, a stable tertiary carbocation must have been formed during the mechanism. Deuterium is an isotope of hydrogen having similar chemical properties. When an alkene is treated with deuterium bromide, the first step of addition is that the deuterium gets attached to the doubly bonded carbon atom which has got a higher number of hydrogens attached (or least substituted). Thus, the carbon atom to which deuterium is attached must be having a double bond in the original alkene. The resulting carbocation that would form will be secondary and can rearrange to a more stable, tertiary carbocation via 1, 2-hydride shift. Thus, a less stable carbocation undergoes carbocation rearrangement reaction to form the most stable carbocation. In the next step, the bromine ion attacks this tertiary carbocation to form the given product.
Thus, the structure of the corresponding alkene that could have been used to produce the given deuterated bromoalkane must be
The complete mechanism is shown below:
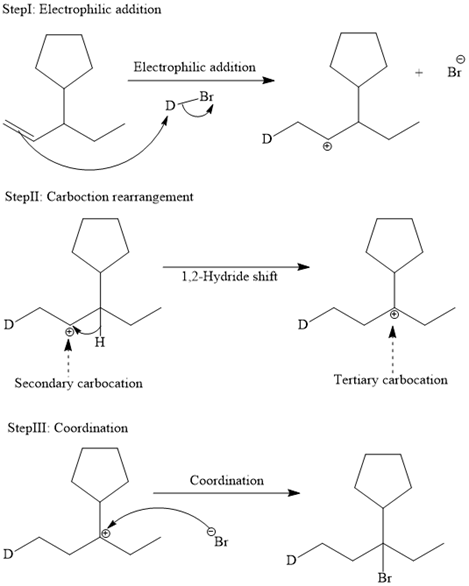
In an electrophilic addition reaction, the carbocation rearrangement reaction takes place to form a rearranged product.
(c)
Interpretation:
The structure of the alkene that could have been used to produce deuterated bromoalkene when treated with deuterium bromide (
Concept introduction:
The electrophilic addition of a Bronsted acid to the carbon-carbon double bond in alkenes is susceptible to carbocation rearrangements due to the stability of the carbocation. The carbocation rearrangement occurs via either 1, 2 hydride shift or 1, 2 methyl shift depending on the stability of the carbocation formed. The stability order for carbocation is benzylic > tertiary > secondary > primary > methyl etc. The normal electrophilic addition gives 1, 2-addition product, but due to the rearrangement reaction, the rearranged product may be formed instead of 1, 2-addition product. In the reaction of an alkene and hydrogen halide or deuterium halide, the carbon to which the deuterium is attached is the carbon bearing the positive charge. Further, the carbon atom to which the halide atom is attached must be the carbon bearing the positive charge, that is, carbocation intermediate. Rearrangements of the carbocation intermediate may be observed if a more stable carbocation is possible.
Answer to Problem 11.8P
The structure of the corresponding alkene that has been used to produce the given deuterated bromoalkane is
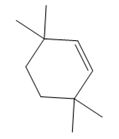
Explanation of Solution
The structure of the given bromoalkane is
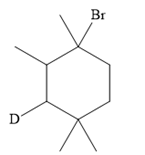
In the structure above, the carbon atom to which the bromine atom is attached must be the carbon bearing the positive charge. Thus, a stable tertiary carbocation must have been formed during the mechanism. Deuterium is an isotope of hydrogen having similar chemical properties. When an alkene is treated with deuterium bromide, the first step of addition is that the deuterium gets attached to the doubly bonded carbon atom, which has got a higher number of hydrogens attached (or least substituted). Thus, the carbon atom to which deuterium is attached must be having a double bond in the original alkene. The resulting carbocation that would form will be secondary and can rearrange to a more stable, tertiary carbocation via 1, 2-hydride shift. Thus, a less stable carbocation undergoes carbocation rearrangement reaction to form the most stable carbocation. In the next step, the bromine ion attacks this tertiary carbocation to form the given product.
Thus, the structure of the corresponding alkene that has been used to produce the given deuterated bromoalkane is
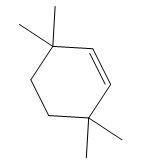
The complete mechanism is shown below:
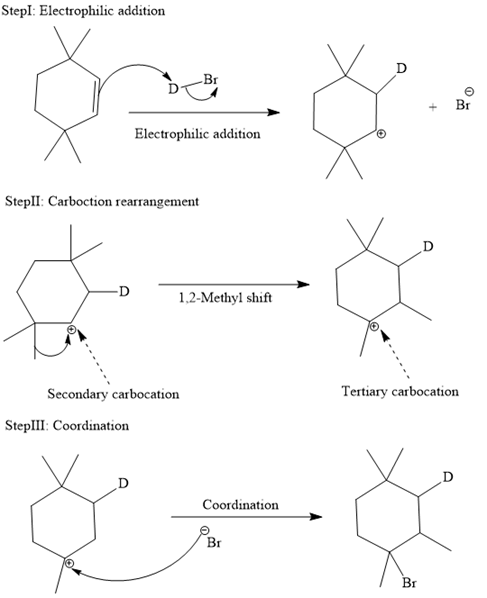
In an electrophilic addition reaction, the carbocation rearrangement reaction takes place to form a rearranged product.
(d)
Interpretation:
The structure of the alkene that could have been used to produce deuterated bromoalkene when treated with deuterium bromide (
Concept introduction:
The electrophilic addition of a Bronsted acid to the carbon-carbon double bond in alkenes is susceptible to carbocation rearrangements due to the stability of the carbocation. The carbocation rearrangement occurs via either 1, 2 hydride shift or 1, 2 methyl shift depending on the stability of the carbocation formed. The stability order for carbocation is benzylic > tertiary > secondary > primary > methyl etc. The normal electrophilic addition gives 1, 2-addition product, but due to the rearrangement reaction, the rearranged product may be formed instead of 1, 2-addition product. In the reaction of an alkene and hydrogen halide or deuterium halide, the carbon to which the deuterium is attached is the carbon bearing the positive charge. Further, the carbon atom to which the halide atom is attached must be the carbon bearing the positive charge, that is, carbocation intermediate. Rearrangements of the carbocation intermediate may be observed if a more stable carbocation is possible.
Answer to Problem 11.8P
The structure of the corresponding alkene that has been used to produce the given deuterated bromoalkane is
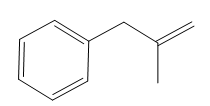
Explanation of Solution
The structure of the given bromoalkane is
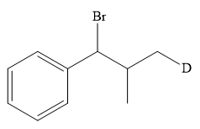
In the structure above, the carbon atom to which the bromine atom is attached must be the carbon bearing the positive charge. Thus, a stable tertiary carbocation must have been formed during the mechanism. Deuterium is an isotope of hydrogen having similar chemical properties. When an alkene is treated with deuterium bromide, the first step of addition is that the deuterium gets attached to the doubly bonded carbon atom, which has got a higher number of hydrogens attached (or least substituted). Thus, the carbon atom to which deuterium is attached must be having a double bond in the original alkene. The resulting carbocation that would form will be secondary and can rearrange to a more stable, tertiary carbocation via 1, 2-hydride shift. Thus, a less stable carbocation undergoes carbocation rearrangement reaction to form the most stable carbocation. In the next step, the bromine ion attacks this tertiary carbocation to form the given product.
Thus, the structure of the corresponding alkene that has been used to produce the given deuterated bromoalkane is
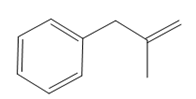
The mechanism for the reaction is shown below:
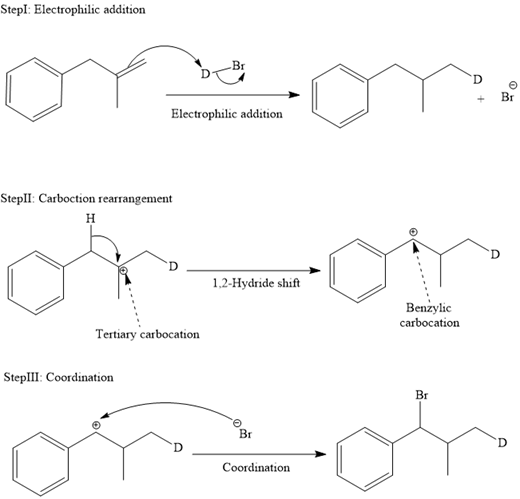
In an electrophilic addition reaction, the carbocation rearrangement reaction takes place to form a rearranged product.
Want to see more full solutions like this?
Chapter 11 Solutions
ORGANIC CHEM BUNDLE
- Solve correctly please, with detailed explaination. When solving alkene addition reactions, how do you know which double bond undergoes the addition reaction if there is more than one? (Gpt/Ai wrong answer not allowed)arrow_forwardThis is a hydrogenation and carbene addition problem. How do I draw the structure of the major organic product of the reaction shown?arrow_forward(SYN) Each of the following compounds can be produced from an alkene, using a single electrophilic addition reaction.Write that reaction and draw its complete, detailed mechanism. (a) 4-chloro-1,2-dimethylcyclohexane; (b) 1-chloro-1,2-dimethylcyclohexane; (c) 1-bromo-1,1-diphenylbutane; (d) 2,2-dichloropentanearrow_forward
- From the curved arrows drawn, determine the products of each of the following reactions.arrow_forwardDraw all of the possible alkene products from the following reaction. Indicate in what amounts alkenes would be produced relative to one another.arrow_forwardWhich of the following addition reactions is not likely to occur? Why? (Please explain clearly, have already done problem)arrow_forward
- . Use to curved arrow notation, propose a mechanism for the following reaction and state whether it is either SN1, E1, SN2, or E2. Give the IUPAC names of all organic reactants and products.arrow_forwardWhich of the following is true? a. Both alkane and alkenes can undergo radical mechanized reaction but mostly for alkanes. b. All reactions of alkenes and alkynes are classified as electrophilic addition reaction. c. Both alkane and alkenes can undergo oxidation reactions to form various oxidation product. d. None of the above.arrow_forwardPredict/draw the major product of the reaction shown in the picture:arrow_forward
- (SYN) Draw the diene that would react with ethene to produce each of the following compoundsarrow_forwardwhich of the following is product of this reaction?arrow_forwardDraw a mechanism for the reaction of methylamine with formic acid. In the box to the left, draw any necessary curved arrows. Show the products of the reaction in the box to the right. Include any nonzero formal charges and all lone pairs of electrons. Finally, check the box to indicate which side of the reaction is favored at equilibrium.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





