
Concept explainers
Write structural formulas for each of the following:
Interpretation:
The structural formula of the given compounds is to be written.
Concept introduction:
When writing the structural formula of any compound, first the functional group from the suffix of the given name is identified.
The longest carbon chain containing the functional group is located.
The carbon atoms of the chain are numbered in a way that the functional group is at lowest numbered carbon atom.
Substituents are attached to the parent chain according to their positions given in the name.
In alkenes, the Z isomers have the higher ranked substituents on the same side of the double bond, and in E isomers, higher ranked substituents are on the opposite sides of the double bond.
Answer to Problem 26P
Solution:
a)
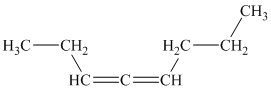
b)
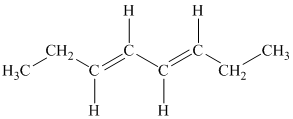
c)
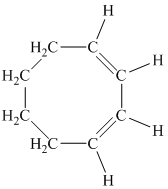
d)
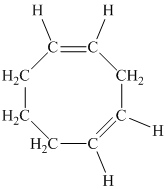
e)
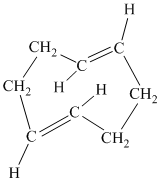
f)
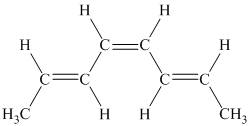
g)
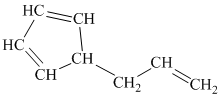
h)
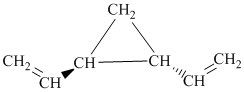
i)
Explanation of Solution
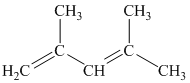
a) The given compound is
The parent name contains the word “
The structural formula is as follows:

b) The given compound is
The parent name contains the word “
The structural formula is as follows:
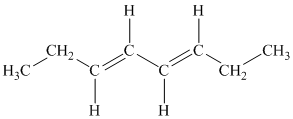
c) The given compound is
The parent name contains the word “cycloocta” in it, which means that the parent carbon chain contains eight carbon atoms in a cyclic form. The suffix added is
The structural formula is as follows:
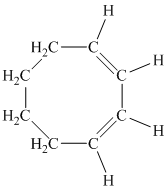
d) The given compound is
The parent name contains the word “cycloocta” in it, which means that the parent carbon chain contains eight carbon atoms in a cyclic form. The suffix added is
The structural formula is as follows:
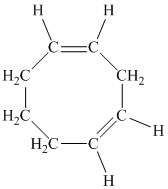
e) The given compound is
The parent name contains the word “cycloocta” in it, which means that the parent carbon chain contains eight carbon atoms in a cyclic form. The suffix added is
The structural formula is as follows:
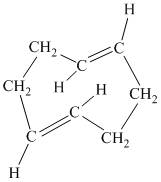
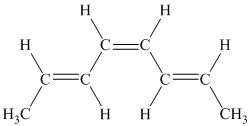
f) The given compound is
The parent name contains the word “
The structural formula is as follows:
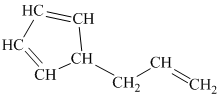
g) The given compound is
The parent name contains the word “cyclopenta” in it, which means that the parent carbon chain contains five carbon atoms in a cyclic form. The suffix added is
The structural formula is as follows:
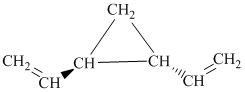
h) The given compound is
The parent name contains the word cyclopropane in it, which means that the parent carbon chain contains three carbon atoms in a cyclic form. There are two vinyl groups attached to the cyclopropane. The numbers
The structural formula is as follows:
i) The given compound is
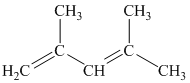
The parent name contains the word “penta” in it, which means that the longest carbon chain contains five carbon atoms. The suffix added is
The structural formula is as follows:
Want to see more full solutions like this?
Chapter 11 Solutions
ORGANIC CHEMISTRY (LL)-W/SOLN.>CUSTOM<
- 12-77 Show how to convert cyclopentene into these compounds. 1,2-Dibromocyclopentane Cyclopentanol Iodocyclopentane Cyclopentanearrow_forwardAddition of HBr to 1,3-Butadiene produce A- 3-Bromo-1-butene B- 1-Bromo-2-butene C- 3,4-dibromo-1-butene D-A and B A O BO C Oarrow_forwardFirst Writedown which reaction it is? SN1, SN2, E1? Write a stepwise mechanism for the following reactions showing ALL intermediates. Use curved arrows to symbolize the flow of electrons to show how each of the intermediates and product are formed. Show all necessary lone pairs and formal charges.arrow_forward
- What is the possible products for following compound with one equivalent of HBr? 3,4 dimethyl-hepta-2,4-diene a) 3 bromo-4,5-dimethyl hepta-3-ene and 4- bromo-3,4-dimethyl hepta-2- ene b) 3 bromo-3,4-dimethyl hepta-3-ene and 4- bromo-3,4-dimethyl hepta-3- ene c) 3 bromo-4,5-dimethyl hepta-2-ene and 3- bromo-3,4-dimethyl hepta-2- ene d) 3 bromo-4,5-dimethyl hepta-3-ene and 4- bromo-3,4-dimethyl hepta-3- ene e) 3 bromo-4,5-dimethyl hepta-3-ene and 2- bromo-3,4-dimethyl hepta-2- ene O a. e O b. b O c.d O d. c O e. aarrow_forwardPlease don't provide handwriting solutionarrow_forwardPlease explain as you provide the correct answers. Thank you.arrow_forward
- Name the alkenes below. Use ONLY E/Z designators to indicate stereochemistry. H3C H Ja H CH₂-C H CH3 CH₂CH3arrow_forwardwhat are the structures of the five alkenes that can produce 3-bromo-3-methylhexane on reaction with HBr 2-ethyl-pentene (E)-3-methylhex-2-ene (Z)-3-methylhex-2-ene (E)-3-methylhex-3-ene (Z)-3-methylhex-3-enearrow_forwardThe products created when 1-methylcyclopentene reacts with one equivalence hydrogen bromide are: H3C Br CH3 CH3 Br 1 2 3 Br Br CH3 CH, Br Br Br 6 5 Br 4,arrow_forward
- What are the starting reactants for the following reaction? 4 X = cyclohexa-1,3-diene; Y = ethyl acrylate X = cyclopenta-1,3-diene; Y = acrylaldehyde X = cyclohexa-1,3-ene; Y = ethyl acrylate X = hexa-1,3-diene; Y = ethyl propiolate CO₂C₂H5arrow_forwardName (including E/Z stereochemistry) the five alkenes that can produce 3-bromo-3-methylhexane on reaction with HBr. Draw the skeletal structure of each molecule.arrow_forwardUsing the table of bond energies estimate the heat of combustion of one mole of 2,4-dimethyl-1,3-pentadiene.arrow_forward

 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning

