
ORGANIC CHEMISTRY-PACKAGE
8th Edition
ISBN: 9780134647586
Author: Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 11, Problem 38P
Using the given starting material, any necessary inorganic reagent and catalysts, and any carbon-containing compound with no more than two carbons, indicate how each of the following compounds can be prepared:
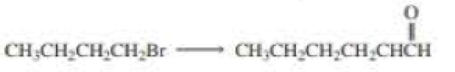
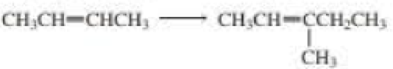
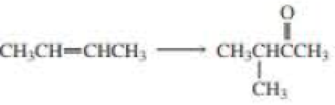
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Using the given starting material, any necessary inorganic reagents and catalysts, and any carbon-containing compounds with no more than three carbons, indicate how each of the following compounds can be prepared:
Using the given starting material, any necessary inorganic reagents, and any carbon-containing compounds with no more than two carbons, indicate how each of the following compounds can be prepared:
What reagents should be used to prepare the following compound
Chapter 11 Solutions
ORGANIC CHEMISTRY-PACKAGE
Ch. 11.1 - Prob. 1PCh. 11.2 - Which is more reactive an organolithium compound...Ch. 11.2 - Prob. 3PCh. 11.3 - Muscalure is the sex attractant of the common...Ch. 11.3 - Prob. 7PCh. 11.3 - Prob. 8PCh. 11.3 - Prob. 9PCh. 11.3 - Prob. 10PCh. 11.4 - Prob. 13PCh. 11.4 - Prob. 14P
Ch. 11.4 - Prob. 15PCh. 11.4 - Prob. 16PCh. 11.4 - Prob. 17PCh. 11.4 - Prob. 19PCh. 11.4 - Show how the Suzuki and/or Heck reactions can be...Ch. 11.4 - Identify two pairs of an alkyl bromide and an...Ch. 11.5 - Prob. 22PCh. 11.5 - Draw the product of ring-closing metathesis for...Ch. 11.5 - Prob. 25PCh. 11.5 - Prob. 26PCh. 11 - Prob. 27PCh. 11 - Prob. 28PCh. 11 - The coupling of an alkyne with an aryl halide in...Ch. 11 - Identify A through H.Ch. 11 - Using the given starting material, any necessary...Ch. 11 - What alkyl halide reacts with lithium...Ch. 11 - Prob. 33PCh. 11 - Prob. 34PCh. 11 - The following compound undergoes an intramolecular...Ch. 11 - Using ethynyleyclohexane as a starting material...Ch. 11 - Prob. 37PCh. 11 - Using the given starting material, any necessary...Ch. 11 - Prob. 39PCh. 11 - A student added an equivalent of...Ch. 11 - Using the given starting material, any necessary...Ch. 11 - Prob. 42PCh. 11 - Prob. 43PCh. 11 - Bombykol is the sex pheromone of the silk moth....Ch. 11 - Prob. 45PCh. 11 - Prob. 46PCh. 11 - A dibromide loses only one bromine when it reacts...Ch. 11 - What starting material is required in order to...Ch. 11 - What product is obtained from ring-opening...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Indicate how the following compound can be synthesized from the given starting material and any other necessary reagent:arrow_forwardIdentify the correct reagent for each of the following reactions.arrow_forwardUsing the given starting material and any necessary organic or inorganic reagents, indicate how the desired compounds could be synthesized:arrow_forward
- What reagents should be used to prepare the following compoundsarrow_forwardUsing the given starting material, any necessary inorganic reagents, and any carbon-containing compounds with no more than two carbons, indicate how the following syntheses could be carried out:arrow_forwardHow should each of the following contents be prepared? Use the specified starting material and any other reagentsarrow_forward
- Using cyclohexanone as the starting material, describe how each of the following compounds can be synthesized:arrow_forwardDescribe how the following compounds can be prepared from compounds containing no more than six carbons.arrow_forwardWhat is the product formed when 1-ethynylcyclohexane is reacted using mercury (II) acid catalyzed hydration?arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you

How to Design a Total Synthesis; Author: Chemistry Unleashed;https://www.youtube.com/watch?v=9jRfAJJO7mM;License: Standard YouTube License, CC-BY