
ORGANIC CHEMISTRY-PACKAGE
8th Edition
ISBN: 9780134647586
Author: Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 11, Problem 48P
What starting material is required in order to synthesize each of the following compounds by ring-closing metathesis?
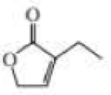
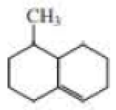
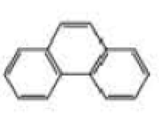
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
What is the major organic product obtained from the following sequence of reactions?
What two sets of reagents (each consisting of a carbonyl compound and phosphonium ylide) can be used for the synthesis of each of the following alkenes?
What alkyl halide is required to prepare each of the phosphonium ylides? What is the best set of reagents to use for the synthesis?
Which of the following will give two identical products upon ozonolysis?
Chapter 11 Solutions
ORGANIC CHEMISTRY-PACKAGE
Ch. 11.1 - Prob. 1PCh. 11.2 - Which is more reactive an organolithium compound...Ch. 11.2 - Prob. 3PCh. 11.3 - Muscalure is the sex attractant of the common...Ch. 11.3 - Prob. 7PCh. 11.3 - Prob. 8PCh. 11.3 - Prob. 9PCh. 11.3 - Prob. 10PCh. 11.4 - Prob. 13PCh. 11.4 - Prob. 14P
Ch. 11.4 - Prob. 15PCh. 11.4 - Prob. 16PCh. 11.4 - Prob. 17PCh. 11.4 - Prob. 19PCh. 11.4 - Show how the Suzuki and/or Heck reactions can be...Ch. 11.4 - Identify two pairs of an alkyl bromide and an...Ch. 11.5 - Prob. 22PCh. 11.5 - Draw the product of ring-closing metathesis for...Ch. 11.5 - Prob. 25PCh. 11.5 - Prob. 26PCh. 11 - Prob. 27PCh. 11 - Prob. 28PCh. 11 - The coupling of an alkyne with an aryl halide in...Ch. 11 - Identify A through H.Ch. 11 - Using the given starting material, any necessary...Ch. 11 - What alkyl halide reacts with lithium...Ch. 11 - Prob. 33PCh. 11 - Prob. 34PCh. 11 - The following compound undergoes an intramolecular...Ch. 11 - Using ethynyleyclohexane as a starting material...Ch. 11 - Prob. 37PCh. 11 - Using the given starting material, any necessary...Ch. 11 - Prob. 39PCh. 11 - A student added an equivalent of...Ch. 11 - Using the given starting material, any necessary...Ch. 11 - Prob. 42PCh. 11 - Prob. 43PCh. 11 - Bombykol is the sex pheromone of the silk moth....Ch. 11 - Prob. 45PCh. 11 - Prob. 46PCh. 11 - A dibromide loses only one bromine when it reacts...Ch. 11 - What starting material is required in order to...Ch. 11 - What product is obtained from ring-opening...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- What reaction presented in this chapter is occurring in the following equation? Explain the resulting stereochemistry of the reaction.arrow_forwardWhat alkene is required to synthesize the following compound? What other epoxide would be formed?arrow_forwardProvide the reagents necessary to accomplish each of the following transformation.arrow_forward
- What alkene should be used to synthesize each of the following alkyl bromides?arrow_forwardWhat is the expected major product for the following reaction sequence?arrow_forwardWhich of A to E correctly shows the structure of the major organic product obtained from the ozonolysis reaction shown below?arrow_forward
- What two sets of reagents (each consisting of a carbonyl compound and phosphonium ylide) can be used for the synthesis of each of the following alkenes?arrow_forwardWhich of the following is NOT a correct statement about electrophilic aromatic substitution mechanism? Formation of a carbocation intermediate is the rate-determining step. The carbocation intermediate contains an sp3 hybridized carbon in the ring. The addition product is a frequent minor product. Aromaticity is regained by loss of H+ Benzene functions as nucleophile.arrow_forwardWhat two sets of reagents (each consisting of a carbonyl compound and phosphonium ylide) can be used for the synthesis of the following alkene? a. What alkyl halide is required to prepare each of the phosphonium ylides? b. What is the best set of reagents to use for the synthesis?arrow_forward
- What carbon-carbon bond forming reaction would you want to consider to synthesize the following compound?arrow_forwardWhich of the following is most susceptible towards nucleophilic aromatic substitution?arrow_forwardSupply the missing reagent in the following reaction: (see attachment) A. Lindlar, H2, mCPBA B. O3 or KMnO4 C. fused KOH D. NH3 E. NaNH2arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
Characteristic Reactions of Benzene and Phenols; Author: Linda Hanson;https://www.youtube.com/watch?v=tjEqEjDd87E;License: Standard YouTube License, CC-BY
An Overview of Aldehydes and Ketones: Crash Course Organic Chemistry #27; Author: Crash Course;https://www.youtube.com/watch?v=-fBPX-4kFlw;License: Standard Youtube License