![OWLv2 for Moore/Stanitski's Chemistry: The Molecular Science, 5th Edition, [Instant Access], 1 term (6 months)](https://s3.amazonaws.com/compass-isbn-assets/textbook_empty_images/large_textbook_empty.svg)
OWLv2 for Moore/Stanitski's Chemistry: The Molecular Science, 5th Edition, [Instant Access], 1 term (6 months)
5th Edition
ISBN: 9781285460420
Author: John W. Moore; Conrad L. Stanitski
Publisher: Cengage Learning US
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 11, Problem 82QRT
The reaction
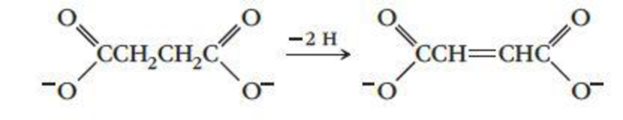
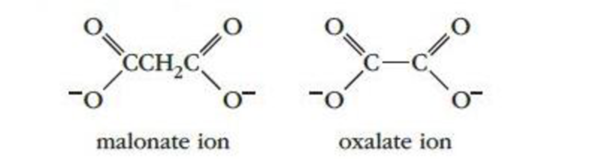
is catalyzed by the enzyme succinate dehydrogenase. When malonate ions or oxalate ions are added to the reaction mixture, the rate decreases significantly. Try to account for this observation in terms of the description of enzyme catalysis given in the text. The structures of malonate and oxalate ions are
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Chapter 11 Solutions
OWLv2 for Moore/Stanitski's Chemistry: The Molecular Science, 5th Edition, [Instant Access], 1 term (6 months)
Ch. 11.1 - For the reaction of crystal violet with NaOH(aq),...Ch. 11.1 - (a) From data in Table 11.1, calculate the rate of...Ch. 11.1 - For the reaction 4NO2(g)+O2(g)2N2O5(g) (a) express...Ch. 11.1 - Instantaneous rates for the reaction of hydroxide...Ch. 11.1 - Prob. 11.3CECh. 11.2 - Prob. 11.4ECh. 11.2 - Prob. 11.3PSPCh. 11.2 - Prob. 11.5ECh. 11.3 - Prob. 11.4PSPCh. 11.3 - Prob. 11.5PSP
Ch. 11.3 - Prob. 11.6PSPCh. 11.3 - Prob. 11.7PSPCh. 11.4 - Prob. 11.6ECh. 11.4 - Prob. 11.7CECh. 11.4 - Prob. 11.8PSPCh. 11.4 - Prob. 11.8CECh. 11.5 - Prob. 11.9PSPCh. 11.5 - The frequency factor A is 6.31 108 L mol1 s1 and...Ch. 11.6 - Prob. 11.10CECh. 11.7 - Prob. 11.11ECh. 11.7 - The Raschig reaction produces the industrially...Ch. 11.7 - Prob. 11.12ECh. 11.8 - The oxidation of thallium(I) ion by cerium(IV) ion...Ch. 11.9 - Prob. 11.11PSPCh. 11.9 - Prob. 11.14CECh. 11 - An excellent way to make highly pure nickel metal...Ch. 11 - Prob. 1QRTCh. 11 - Prob. 2QRTCh. 11 - Prob. 3QRTCh. 11 - Prob. 4QRTCh. 11 - Prob. 5QRTCh. 11 - Prob. 6QRTCh. 11 - Prob. 7QRTCh. 11 - Prob. 8QRTCh. 11 - Prob. 9QRTCh. 11 - Prob. 10QRTCh. 11 - Prob. 11QRTCh. 11 - Cyclobutane can decompose to form ethylene:
The...Ch. 11 - Prob. 13QRTCh. 11 - Prob. 14QRTCh. 11 - For the reaction 2NO2(g)2NO(g)+O2(g) make...Ch. 11 - Prob. 16QRTCh. 11 - Prob. 17QRTCh. 11 - Ammonia is produced by the reaction between...Ch. 11 - Prob. 19QRTCh. 11 - Prob. 20QRTCh. 11 - The reaction of CO(g) + NO2(g) is second-order in...Ch. 11 - Nitrosyl bromide, NOBr, is formed from NO and Br2....Ch. 11 - Prob. 23QRTCh. 11 - Prob. 24QRTCh. 11 - Prob. 25QRTCh. 11 - For the reaction
these data were obtained at 1100...Ch. 11 - Prob. 27QRTCh. 11 - Prob. 28QRTCh. 11 - Prob. 29QRTCh. 11 - Prob. 30QRTCh. 11 - Prob. 31QRTCh. 11 - Prob. 32QRTCh. 11 - For the reaction of phenyl acetate with water the...Ch. 11 - When phenacyl bromide and pyridine are both...Ch. 11 - The compound p-methoxybenzonitrile N-oxide, which...Ch. 11 - Prob. 36QRTCh. 11 - Radioactive gold-198 is used in the diagnosis of...Ch. 11 - Prob. 38QRTCh. 11 - Prob. 39QRTCh. 11 - Prob. 40QRTCh. 11 - Prob. 41QRTCh. 11 - Prob. 42QRTCh. 11 - Prob. 43QRTCh. 11 - Prob. 44QRTCh. 11 - Prob. 45QRTCh. 11 - Prob. 46QRTCh. 11 - Prob. 47QRTCh. 11 - Prob. 48QRTCh. 11 - Prob. 49QRTCh. 11 - Prob. 50QRTCh. 11 - Prob. 51QRTCh. 11 - Prob. 52QRTCh. 11 - For the reaction of iodine atoms with hydrogen...Ch. 11 - Prob. 54QRTCh. 11 - The activation energy Ea is 139.7 kJ mol1 for the...Ch. 11 - Prob. 56QRTCh. 11 - Prob. 57QRTCh. 11 - Prob. 58QRTCh. 11 - Prob. 59QRTCh. 11 - Prob. 60QRTCh. 11 - Prob. 61QRTCh. 11 - Prob. 62QRTCh. 11 - Prob. 63QRTCh. 11 - Which of the reactions in Question 62 would (a)...Ch. 11 - Prob. 65QRTCh. 11 - Prob. 66QRTCh. 11 - Prob. 67QRTCh. 11 - Prob. 68QRTCh. 11 - Prob. 69QRTCh. 11 - Prob. 70QRTCh. 11 - Prob. 71QRTCh. 11 - For the reaction the rate law is Rate=k[(CH3)3CBr]...Ch. 11 - Prob. 73QRTCh. 11 - Prob. 74QRTCh. 11 - Prob. 75QRTCh. 11 - For this reaction mechanism,
write the chemical...Ch. 11 - Prob. 77QRTCh. 11 - Prob. 78QRTCh. 11 - Prob. 79QRTCh. 11 - When enzymes are present at very low...Ch. 11 - Prob. 81QRTCh. 11 - The reaction is catalyzed by the enzyme succinate...Ch. 11 - Prob. 83QRTCh. 11 - Many biochemical reactions are catalyzed by acids....Ch. 11 - Prob. 85QRTCh. 11 - Prob. 86QRTCh. 11 - Prob. 87QRTCh. 11 - Prob. 88QRTCh. 11 - Prob. 89QRTCh. 11 - Prob. 90QRTCh. 11 - Prob. 91QRTCh. 11 - Prob. 92QRTCh. 11 - Prob. 93QRTCh. 11 - Prob. 94QRTCh. 11 - Nitryl fluoride is an explosive compound that can...Ch. 11 - Prob. 96QRTCh. 11 - Prob. 97QRTCh. 11 - For a reaction involving the decomposition of a...Ch. 11 - Prob. 99QRTCh. 11 - Prob. 100QRTCh. 11 - Prob. 101QRTCh. 11 - This graph shows the change in concentration as a...Ch. 11 - Prob. 103QRTCh. 11 - Prob. 104QRTCh. 11 - Prob. 105QRTCh. 11 - Prob. 106QRTCh. 11 - Prob. 107QRTCh. 11 - Prob. 108QRTCh. 11 - Prob. 109QRTCh. 11 - Prob. 110QRTCh. 11 - Prob. 111QRTCh. 11 - Prob. 112QRTCh. 11 - Prob. 113QRTCh. 11 - Prob. 114QRTCh. 11 - Prob. 115QRTCh. 11 - Prob. 116QRTCh. 11 - Prob. 118QRTCh. 11 - Prob. 119QRTCh. 11 - In a time-resolved picosecond spectroscopy...Ch. 11 - If you know some calculus, derive the integrated...Ch. 11 - If you know some calculus, derive the integrated...Ch. 11 - (Section 11-5) A rule of thumb is that for a...Ch. 11 - Prob. 11.BCPCh. 11 - Prob. 11.CCPCh. 11 - Prob. 11.DCP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- In each of the following, which reaction mechanism assumption is apparently being violated? Explain your answers. a. A reaction takes place more rapidly when the concentration of reactants is decreased. b. A reaction takes place more rapidly when the reaction mixture is cooled. c. The reaction rate of A+BAB increases as the concentration of A is increased but does not change as the concentration of B is increased.arrow_forwardWhy are enzymes important? For example, what is the importance of the enzyme carbonic anhydrase in the body?arrow_forwardDescribe the difference between graphs showing temperature versus reaction rate for an enzyme-catalyzed reaction and an uncatalyzed reaction.arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning


Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Introductory Chemistry: An Active Learning Approa...
Chemistry
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning
Enzymes - Effect of cofactors on enzyme; Author: Tutorials Point (India) Ltd;https://www.youtube.com/watch?v=AkAbIwxyUs4;License: Standard YouTube License, CC-BY
Enzyme Catalysis Part-I; Author: NPTEL-NOC IITM;https://www.youtube.com/watch?v=aZE740JWZuQ;License: Standard Youtube License