
Study Guide for Zumdahl/Zumdahl/DeCoste’s Chemistry, 10th Edition
10th Edition
ISBN: 9781305957473
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 11, Problem 97AE
The solubility of benzoic acid (HC7H5O2),
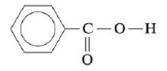
is 0.34 g/100 mL in water at 25°C and is 10.0 g/100 mL in benzene (C6H6) at 25°C. Rationalize this solubility behavior. (Hint: Benzoic acid forms a dimer in benzene.) Would benzoic acid be more or less soluble in a 0.1-M NaOH solution than it is in water? Explain.
Expert Solution & Answer
Trending nowThis is a popular solution!

Chapter 11 Solutions
Study Guide for Zumdahl/Zumdahl/DeCoste’s Chemistry, 10th Edition
Ch. 11 - Prob. 1RQCh. 11 - Using KF as an example, write equations that refer...Ch. 11 - Prob. 3RQCh. 11 - Prob. 4RQCh. 11 - Define the terms in Raoults law. Figure 10-9...Ch. 11 - In terms of Raoults law, distinguish between an...Ch. 11 - Vapor-pressure lowering is a colligative property,...Ch. 11 - What is osmotic pressure? How is osmotic pressure...Ch. 11 - Distinguish between a strong electrolyte, a weak...Ch. 11 - Prob. 10RQ
Ch. 11 - Prob. 1ALQCh. 11 - Once again, consider Fig. 10-9. Suppose instead of...Ch. 11 - Prob. 3ALQCh. 11 - Prob. 4ALQCh. 11 - You have read that adding a solute to a solvent...Ch. 11 - You drop an ice cube (made from pure water) into a...Ch. 11 - Using the phase diagram for water and Raoults law,...Ch. 11 - You and your friend are each drinking cola from...Ch. 11 - Prob. 9ALQCh. 11 - Prob. 10ALQCh. 11 - If a solution shows positive deviations from...Ch. 11 - Prob. 12ALQCh. 11 - Rubbing alcohol contains 585 g isopropanol...Ch. 11 - Prob. 14SRCh. 11 - Prob. 15SRCh. 11 - Prob. 16SRCh. 11 - Calculate the sodium ion concentration when 70.0...Ch. 11 - Write equations showing the ions present after the...Ch. 11 - Rationalize the temperature dependence of the...Ch. 11 - The weak electrolyte NH3(g) does not obey Henrys...Ch. 11 - The two beakers in the sealed container...Ch. 11 - The following plot shows the vapor pressure of...Ch. 11 - When pure methanol is mixed with water, the...Ch. 11 - Prob. 24QCh. 11 - For an acid or a base, when is the normality of a...Ch. 11 - Prob. 26QCh. 11 - Prob. 27QCh. 11 - Prob. 28QCh. 11 - Prob. 29QCh. 11 - Table sugar (C12H22O11) or urea [(NH2)2CO] can be...Ch. 11 - If two different aqueous solutions of proteins...Ch. 11 - An extremely important application of dialysis is...Ch. 11 - Explain the terms isotonic solution, crenation,...Ch. 11 - Prob. 34QCh. 11 - Prob. 35ECh. 11 - A typical IV used in hospitals is dextrose 5% in...Ch. 11 - Prob. 37ECh. 11 - Prob. 38ECh. 11 - Common commercial acids and bases are aqueous...Ch. 11 - In lab you need to prepare at least 100 mL of each...Ch. 11 - Prob. 41ECh. 11 - Prob. 42ECh. 11 - Prob. 43ECh. 11 - Calculate the molarity and mole fraction of...Ch. 11 - Prob. 45ECh. 11 - Prob. 46ECh. 11 - Prob. 47ECh. 11 - a. Use the following data to calculate the...Ch. 11 - Although Al(OH)3 is insoluble in water, NaOH is...Ch. 11 - Prob. 50ECh. 11 - Prob. 51ECh. 11 - Prob. 52ECh. 11 - For each of the following pairs, predict which...Ch. 11 - Which ion in each of the following pairs would you...Ch. 11 - Rationalize the trend in water solubility for the...Ch. 11 - In flushing and cleaning columns used in liquid...Ch. 11 - The solubility of nitrogen in water is 8.21 104...Ch. 11 - Calculate the solubility of O2 in water at a...Ch. 11 - The vapor pressure of a solution containing 53.6 g...Ch. 11 - An aqueous solution containing glucose has a vapor...Ch. 11 - The normal boiling point of diethyl ether is...Ch. 11 - At a certain temperature, the vapor pressure of...Ch. 11 - Prob. 63ECh. 11 - A solution is prepared by mixing 0.0300 mole of...Ch. 11 - What is the composition of a methanol...Ch. 11 - Benzene and toluene form an ideal solution....Ch. 11 - Which of the following will have the lowest total...Ch. 11 - Prob. 68ECh. 11 - Match the vapor pressure diagrams with the...Ch. 11 - The vapor pressures of several solutions of...Ch. 11 - A solution is prepared by dissolving 27.0 g urea,...Ch. 11 - A 2.00-g sample of a large biomolecule was...Ch. 11 - What mass of glycerin (C3H8O3), a nonelectrolyte,...Ch. 11 - The freezing point of 1-butanol is 25.50C and Kf...Ch. 11 - Prob. 75ECh. 11 - What volume of ethylene glycol (C2H6O2), a...Ch. 11 - Reserpine is a natural product isolated from the...Ch. 11 - A solution contains 3.75 g of a nonvolatile pure...Ch. 11 - a. Calculate the freezing-point depression and...Ch. 11 - Erythrocytes are red blood cells containing...Ch. 11 - Prob. 81ECh. 11 - Prob. 82ECh. 11 - Prob. 83ECh. 11 - Prob. 84ECh. 11 - Consider the following solutions: 0.010 m Na3PO4...Ch. 11 - From the following: pure water solution of...Ch. 11 - Prob. 87ECh. 11 - Prob. 88ECh. 11 - Prob. 89ECh. 11 - Consider the following representations of an ionic...Ch. 11 - Prob. 91ECh. 11 - Prob. 92ECh. 11 - Use the following data for three aqueous solutions...Ch. 11 - The freezing-point depression of a 0.091-m...Ch. 11 - Prob. 95ECh. 11 - A 0.500-g sample of a compound is dissolved in...Ch. 11 - The solubility of benzoic acid (HC7H5O2), is 0.34...Ch. 11 - Prob. 99AECh. 11 - Prob. 100AECh. 11 - Prob. 101AECh. 11 - In Exercise 96 in Chapter 8, the pressure of CO2...Ch. 11 - Explain the following on the basis of the behavior...Ch. 11 - The term proof is defined as twice the percent by...Ch. 11 - Prob. 105AECh. 11 - Prob. 106AECh. 11 - A solution is made by mixing 50.0 g acetone...Ch. 11 - Prob. 108AECh. 11 - Thyroxine, an important hormone that controls the...Ch. 11 - Prob. 110AECh. 11 - An unknown compound contains only carbon,...Ch. 11 - Prob. 112AECh. 11 - Prob. 113AECh. 11 - Prob. 115AECh. 11 - Patients undergoing an upper gastrointestinal...Ch. 11 - Prob. 118CWPCh. 11 - The lattice energy of NaCl is 786 kJ/mol, and the...Ch. 11 - For each of the following pairs, predict which...Ch. 11 - The normal boiling point of methanol is 64.7C. A...Ch. 11 - A solution is prepared by mixing 1.000 mole of...Ch. 11 - Prob. 123CWPCh. 11 - A 4.7 102 mg sample of a protein is dissolved in...Ch. 11 - A solid consists of a mixture of NaNO3 and...Ch. 11 - The vapor pressure of pure benzene is 750.0 torr...Ch. 11 - Prob. 127CPCh. 11 - Plants that thrive in salt water must have...Ch. 11 - You make 20.0 g of a sucrose (C12H22O11) and NaCl...Ch. 11 - Prob. 130CPCh. 11 - The vapor in equilibrium with a pentane-hexane...Ch. 11 - A forensic chemist is given a white solid that is...Ch. 11 - A 1.60-g sample of a mixture of naphthalene...Ch. 11 - Prob. 134CPCh. 11 - Prob. 135CPCh. 11 - You have a solution of two volatile liquids, A and...Ch. 11 - In some regions of the southwest United States,...Ch. 11 - Specifications for lactated Ringers solution,...Ch. 11 - Creatinine, C4H7N3O, is a by-product of muscle...Ch. 11 - An aqueous solution containing 0.250 mole of Q, a...Ch. 11 - Anthraquinone contains only carbon, hydrogen, and...
Additional Science Textbook Solutions
Find more solutions based on key concepts
Determine the de Brogue wavelength of a. an electron moving at 1/10 the speed of light. b. a 400 g Frisbee movi...
Inorganic Chemistry
141. Design a device that uses as electrochemical cell to determine amount of
in a sample water Describe, in...
Chemistry: Structure and Properties (2nd Edition)
Practice Problem 1.22 Which of the following alkenes can exist as cis-trans isomers? Write their structures. Bu...
Organic Chemistry
Q2. Which statement best defines chemistry?
a. The science that studies solvents, drugs, and insecticides
b. Th...
Introductory Chemistry (5th Edition) (Standalone Book)
The method to determine the volume of a powered solid, liquid and a rock needs to be determined. Concept introd...
Living by Chemistry
What is the pH range for acidic solutions? For basic solutions?
EBK INTRODUCTION TO CHEMISTRY
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Bradykinin is a small peptide (9 amino acids; 1060 g/mol) that lowers blood pressure by causing blood vessels to dilate. What is the osmotic pressure of a solution of this protein at 20 C if 0.033 g of the peptide is dissolved in water to give 50.0 mL of solution?arrow_forwardThe solubility of benzoic acid (HC7H5O2), is 0.34 g/100 mL in water at 25C and is 10.0 g/100 mL in benzene (C6H6) at 25C. Rationalize this solubility behavior. (Hint: Benzoic acid forms a dimer in benzene.) Would benzoic acid be more or less soluble in a 0.1-M NaOH solution than it is in water? Explain.arrow_forwardPredict the relative solubility of each compound in the two solvents, based on the intermolecular attractions. (a) Is potassium iodide more soluble in water or in methylene chloride (CH2Cl2)? (b) Is toluene (C6H5CH3) more soluble in benzene (C6H6) or in water? (c) Is ethylene glycol (C2H4(OH)2) more soluble in hexane (C6H14) or in ethanol (C2H5OH)?arrow_forward
- Butane, C4H10, has been suggested as the refrigerant in household compressors such as those found in air conditioners. (a) To what extent is butane soluble in water? Calculate the butane concentration in water if the pressure of the gas is 0.21 atm. (kH = 0.0011 mol/kgbar at 25 C) (b) If the pressure of butane is increased to 1.0 atm, does the butane concentration increase or decrease?arrow_forwardWhat would be the freezing point of a solution formed by adding 1.0 mole of glucose (a molecular compound) to the following amounts of water? a. 250 g (0.25 kg) b. 500 g (0.500 kg) c. 1000 g (1.000 kg) d. 2000 g (2.000 kg)arrow_forwardA 0.109 mol/kg aqueous solution of formic acid, HCOOH, freezes at −0.210 °C. Calculate the percent dissociation of formic acid.arrow_forward
- A solution is made by dissolving 0.455 g of PbBr2 in 100 g of H2O at 50C. Based on the data in Table 8-1, should this solution be characterized as a. saturated or unsaturated b. dilute or concentratedarrow_forwardDefine the terms in Raoults law. Figure 10-9 illustrates the net transfer of water molecules from pure water to an aqueous solution of a nonvolatile solute. Explain why eventually all of the water from the beaker of pure water will transfer to the aqueous solution. If the experiment illustrated in Fig. 10-9 was performed using a volatile solute, what would happen? How do you calculate the total vapor pressure when both the solute and solvent are volatile?arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning


General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning
Solutions: Crash Course Chemistry #27; Author: Crash Course;https://www.youtube.com/watch?v=9h2f1Bjr0p4;License: Standard YouTube License, CC-BY