
Concept explainers
A vertical piston–cylinder device contains a gas at a pressure of 100 kPa. The piston has a mass of 10 kg and a diameter of 14 cm. Pressure of the gas is to be increased by placing some weights on the piston. Determine the local atmospheric pressure and the mass of the weights that will double the pressure of the gas inside the cylinder Answers: 93.6 kPa, 157 kg
The local atmospheric pressure of the vertical piston-cylinder device.
The mass of the weights that will double the pressure of the vertical piston-cylinder device.
Answer to Problem 98RP
The local atmospheric pressure of the vertical piston-cylinder device is
The mass of the weights that will double the pressure of the vertical piston-cylinder device is
Explanation of Solution
Show the free body diagram of the vertical piston-cylinder device.
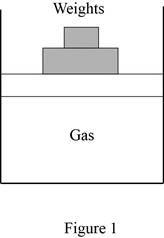
Write the expression of vertical force in the piston-cylinder device.
Here, the mass of piston is
Write the expression of balance force in the piston-cylinder device.
Here, the mass of the weights that will double the pressure is
Determine the area of the piston-cylinder.
Conclusion:
Substitute
Substitute
Thus, the local atmospheric pressure of the vertical piston-cylinder device is
Substitute
Thus, the mass of the weights that will double the pressure of the vertical piston-cylinder device is
Want to see more full solutions like this?
Chapter 1 Solutions
EBK THERMODYNAMICS: AN ENGINEERING APPR
Additional Engineering Textbook Solutions
Engineering Mechanics: Statics & Dynamics (14th Edition)
Fluid Mechanics Fundamentals And Applications
Engineering Mechanics: Statics
Degarmo's Materials And Processes In Manufacturing
Fluid Mechanics: Fundamentals and Applications
Automotive Technology: Principles, Diagnosis, and Service (5th Edition)
- A vertical, frictionless piston-cylinder assembly is filled with gas. The piston has a mass of 3.2 kg and a cross-sectional area of 35 cm². A compressed spring on the piston exerts a force of 150 N on the piston. Determine the pressure in the cylinder in kPa where the atmospheric pressure is 95 kPa.arrow_forwardA vertical, frictionless piston–cylinder device contains a gas at 180 kPa absolute pressure. The atmospheric pressure outside is 100 kPa, and the piston area is 25 cm2 . Determine the mass of the piston.arrow_forwardA vertical piston–cylinder device contains a gas at a pressure of 100 kPa. The piston has a mass of 10 kg and a diameter of 14 cm. Pressure of the gas is to be increased by placing some weights on the piston. Determine the local atmospheric pressure and the mass of the weights that will double the pressure of the gas inside the cylinder.arrow_forward
- A gas is contained in a vertical, frictionless piston– cylinder device. The piston has a mass of 3.2 kg and a cross-sectional area of 35 cm2. A compressed spring above the piston exerts a force of 150 N on the piston. If the atmospheric pressure is 95 kPa, determine the pressure inside the cylinder.arrow_forwardTwo containers each hold 1000 liters of water. The first container is a cube, and the second container is a tall, narrow cylinder. Is the water pressure greater at the bottom of the first container, the second container, or is it the same? Why?arrow_forwardThe piston of a vertical piston-cylinder device containing a gas has a mass of 40 kg and a cross-sectional area of 0.012 m2 . The local atmospheric pressure is 95 kPa, and the gravitational acceleration is 9.81 m/s2 (a) Determine the pressure inside the cylinder. (b) If some heat is transferred to the gas and its volume is doubled, do you expect the pressure inside the cylinder to change?arrow_forward
- A piston-cylinder contains 2 lbm of water. The initial volume is 0.1 ft3. The piston rises, causing the volume to double. Determine the final specific volume of the water.arrow_forwardA vessel of cylindrical shape is 70 cm in diameter and 90 cm high. It contains 7 kg of a gas. The pressure measured with manometer indicates 620 mm of Hg above atmosphere when barometer reads 765 mm of Hg. Determine : (a)The absolute pressure of the gas in the vessel in bar. (b)Specific volume (c) density of the gas.arrow_forwardThe temperature in a pressure cooker during cooking at sea level is measured to be 120°C. Determine the absolute pressure inside the cooker in atm. Would youmodify your answer if the place were at a higher elevation?arrow_forward
- A piston-cylinder assembly contains air at 210 kPa and occupies a volume of 0.01m'. The piston is attached to one end ofa spring and the other end of the spring is fixed to a wall. The force exerted by the spring on the piston is proportional to the decrease in the length of the spring. The ambient atmospheric pressure is 100 kPa. Now, the air in the cylinder is heated till the volume is doubled and at this instant it is found that the pressure of the air in the cylinder is 600 kPa. Sketch the process on p-v diagram and calculate work done by the gas. Q2:arrow_forwardTwo flasks (which we will refer to as flask 1 and flask 2) are connected to each other by a U-shaped tube filled with an oil having a density of 0.564 g mL-1.The oil level in the arm connected to flask 2 is 13.57 cm higher than in the arm connected to flask 1. Flask 1 is also connected to an open-end mercury manometer.The mercury level in the arm open to the atmosphere is 11.70 cm higher than the level in the arm connected to flask 1. The atmospheric pressure is 0.866 atm.What is the pressure of the gas in flask 2 expressed in torr?arrow_forwardA piston-cylinder assembly contains air at 210 kPa and occupies a volume of 0.01m'. The piston is attached to one end of a spring and the other end of the spring is fixed to a wall. The force exerted by the spring on the piston is proportional to the decrease in the length of the spring. The ambient atmospheric pressure is 100 kPa. Now, the air in the cylinder is heated till the volume is doubled and at this instant it is found that the pressure of the air in the cylinder is 600 kPa. Sketch the process on p-v diagram and calculate work done by the gas.arrow_forward
 Refrigeration and Air Conditioning Technology (Mi...Mechanical EngineeringISBN:9781305578296Author:John Tomczyk, Eugene Silberstein, Bill Whitman, Bill JohnsonPublisher:Cengage Learning
Refrigeration and Air Conditioning Technology (Mi...Mechanical EngineeringISBN:9781305578296Author:John Tomczyk, Eugene Silberstein, Bill Whitman, Bill JohnsonPublisher:Cengage Learning
