
Concept explainers
A vertical piston–cylinder device contains a gas at a pressure of 100 kPa. The piston has a mass of 10 kg and a diameter of 14 cm. Pressure of the gas is to be increased by placing some weights on the piston. Determine the local atmospheric pressure and the mass of the weights that will double the pressure of the gas inside the cylinder Answers: 93.6 kPa, 157 kg
The local atmospheric pressure of the vertical piston-cylinder device.
The mass of the weights that will double the pressure of the vertical piston-cylinder device.
Answer to Problem 98RP
The local atmospheric pressure of the vertical piston-cylinder device is
The mass of the weights that will double the pressure of the vertical piston-cylinder device is
Explanation of Solution
Show the free body diagram of the vertical piston-cylinder device.
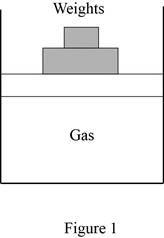
Write the expression of vertical force in the piston-cylinder device.
Here, the mass of piston is
Write the expression of balance force in the piston-cylinder device.
Here, the mass of the weights that will double the pressure is
Determine the area of the piston-cylinder.
Conclusion:
Substitute
Substitute
Thus, the local atmospheric pressure of the vertical piston-cylinder device is
Substitute
Thus, the mass of the weights that will double the pressure of the vertical piston-cylinder device is
Want to see more full solutions like this?
Chapter 1 Solutions
Thermodynamics: An Engineering Approach
Additional Engineering Textbook Solutions
Engineering Mechanics: Statics & Dynamics (14th Edition)
Fluid Mechanics Fundamentals And Applications
Engineering Mechanics: Statics
Degarmo's Materials And Processes In Manufacturing
Fluid Mechanics: Fundamentals and Applications
Automotive Technology: Principles, Diagnosis, and Service (5th Edition)
- A 10-m high cylindrical container with a radius of 2 m is filled with oil and water. If the lower 2.5-m of the tank is water with a density of ρ = 1000 kg/m3 while the rest of the tank is filled with oil with a density of ρ = 800 kg/m3, determine the pressure difference, in kPa, between the top and bottom of the cylinder. Assume that the gravity is 9.81 m/s2.arrow_forwardThe barometer of a mountain hiker reads 980 mbars at the beginning of a hiking trip and 790 mbars at the end. Neglecting the effect of altitude on local gravitational acceleration, determine the vertical distance climbed. Assume an average air density of 1.20 kg/m3arrow_forwardFor a substance, what is the difference between mass and molar mass? How are these two related?arrow_forward
- A vertical, frictionless piston–cylinder device contains a gas at 180 kPa absolute pressure. The atmospheric pressure outside is 100 kPa, and the piston area is 25 cm2 . Determine the mass of the piston.arrow_forwardA U-manometer shown in the figure is initially at rest at the current level of mercury. Determine the maximum amount of water that can be added on the right leg of the manometer in grams if h = 40 cm, DA = 2.5 cm, and DB = 5 cm.arrow_forwardThe helium-filled blimp shown in the figure below is used at various athletic events. Determine the number of pounds of helium (lbm) within it if its volume is 68,000 ft3 and the temperature and pressure are 80 °F and 14.2 psia, respectively. The gas constant for helium is 1.242×104 (ft-lbf)/(slug-R).arrow_forward
- The container is partially filled with oil, water, and air. Determine the pressures at A, B, and C. Take density of water to be 1000 kg/m3, and density of oil to be 830 kg/m3. *The answer should be (A. 22.44 kPa, B. 7.725 kPa,C. 12.63 kPa)*arrow_forwardDuring a picnic on a hot summer day, all the cold drinks disappear quickly, and the only available drinks are those at the ambient temperature of 85°F. In an effort to cool a 12-fluid-oz drink in a can, a person grabs the can and starts shaking it in the iced water of the chest at 32°F. Using the properties of water for the drink, determine the mass of ice that will melt by the time the canned drink cools to 37°F.arrow_forwardDetermine the pressure and specific volume of water at 20 C that has an internal enegy of 1200 KJ/kg.arrow_forward
- The temperature in a pressure cooker during cooking at sea level is measured to be 250°F. Determine the absolute pressure inside the cooker in psia and in atm. Would you modify your answer if the place were at a higher elevation?arrow_forwardA vertical frictionless piston cylinder contains air at a pressure of 300 KPa with atmospheric pressure of 100 KPa. The diameter of the piston is 0.25m, and g = 9.8m/s2. Determine the piston’s mass.arrow_forwardThe gage pressure in a liquid at a depth of 2.5 m is read to be 28 kPa. Determine the gage pressure in the same liquid at a depth of 9 m.arrow_forward
 Refrigeration and Air Conditioning Technology (Mi...Mechanical EngineeringISBN:9781305578296Author:John Tomczyk, Eugene Silberstein, Bill Whitman, Bill JohnsonPublisher:Cengage Learning
Refrigeration and Air Conditioning Technology (Mi...Mechanical EngineeringISBN:9781305578296Author:John Tomczyk, Eugene Silberstein, Bill Whitman, Bill JohnsonPublisher:Cengage Learning
