
Concept explainers
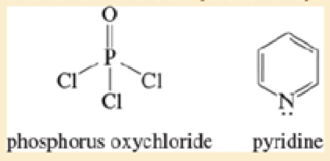
Some alcohols undergo rearrangement or other unwanted side reactions when they dehydrate in acid Alcohols may be dehydrated under mildly basic conditions using phosphorus oxychloride (POCl3) in pyridine. The alcohol reacts with phosphorus oxychloride much like it reacts with tosyl chloride (Section11-5), displacing a chloride ion from phosphorus to give an alkyl dichloroptrosphate ester. The dichlorophosphate group is an outstanding leaving group. Pyridine reacts as a base with the dichlorophosphate ester to give an E2 elimination. Propose a mechanism for the dehydration of cyclohexanol by POCl3 in pyridine.
Want to see the full answer?
Check out a sample textbook solution
Chapter 11 Solutions
ORGANIC CHEMISTRY MASTERINGCHEM ACCESS
Additional Science Textbook Solutions
Chemistry: Structure and Properties
Chemistry
General, Organic, & Biological Chemistry
Organic Chemistry
Chemistry For Changing Times (14th Edition)
Chemistry: An Introduction to General, Organic, and Biological Chemistry (12th Edition) - Standalone book
- Following is the structural formula of the tranquilizer meparfynol (Oblivon). Propose a synthesis for this compound starting with acetylene and a ketone. (Notice the -yn- and -ol in the chemical name of this compound, indicating that it contains alkyne and hydroxyl functional groups.)arrow_forwardClaisen condensation between diethyl phthalate and ethyl acetate followed by saponification, acidification, and decarboxylation forms a diketone, C9H6O2. Propose structural formulas for compounds A and B and the diketone.arrow_forwardAldehydes and ketones react with one molecule of an alcohol to form compounds called hemiacetals, in which there is one hydroxyl group and one ether-like group. Reaction of a hemiacetal with a second molecule of alcohol gives an acetal and a molecule of water. We study this reaction in Chapter 16. Draw structural formulas for the hemiacetal and acetal formed from these reagents. The stoichiometry of each reaction is given in the problem.arrow_forward
- The following sequence of steps converts (R)-2-octanol to (S)-2-octanol. Propose structural formulas for intermediates A and B, specify the configuration of each, and account for the inversion of configuration in this sequence.arrow_forwardOne frequently used method for preparing methyl esters is by reaction of carboxylic acids with diazomethane, CH2N2. The reaction occurs in two steps: (l) protonation of diazomethane by the carboxylic acid to yield methyldiazonium ion, CH3N2+, plus a carboxylate ion; and (2) reaction of the carboxylate ion with CH3N2+. (a) Draw two resonance structures of diazomethane, and account for step 1. (b) What kind of reaction occurs in step 2?arrow_forwardNonconjugated , -unsaturated ketones, such as 3-cyclohexenone, are in an acid-catalyzed equilibrium with their conjugated , -unsaturated isomers. Propose a mechanism for this isomerization.arrow_forward
- There are several isomeric alcohols and ethers of molecular formula C5H12O. Propose a structure for the isomer. Isomer B: δ = 0.92 (t, 7.8 Hz, 3 H), 1.20 (s, 6H), 1.49 (q, 7.8 Hz, 2H), 1.85 (s, 1H) ppmarrow_forward3.) Starting from phenol, the synthesis of 2,4-dichlorophenoxyacetic acid involves four steps outlined by the roadmap below. Please provide the missing reagents and products where necessary. OH 1. NaOH HCl(aq) 2 Cl₂ FeCla OH 2,4-dichlorophenoxyacetic acid Iarrow_forwardDimethyl disulfide, CH,S–SCH3, found in the vaginal secretions of female hamsters, acts as a sexual attractant for the male hamster. Write an equation for its synthesis from methanethiol.arrow_forward
- Predict the products of the following acid-base reactions. If the equilibrium would not result in the formation of appreciable amounts of products, you should so indicate. In each case label the stronger acid, the stronger base, the weaker acid, and the weaker base: (a) CH3CH=CH2 + NANH2 (d) CH3C=C: + CH;CH2OH → (e) CH3C=C:- + NH¾CI – | (b) CH;C=CH + NaNH2 (c) CH3CH2CH3 + NANH2 → | HASarrow_forwardStructure of product C A hydrocarbon, A, with formula C11H16 undergoes hydrogenation with Pd to give product B, C11H22, and with Lindlar's catalyst to give product C, C11 H18. Ozonolysis leads to a mixture of products including cyclohexanone and 4-oxobutanoic acid. Treatment with sodium amide (NaNH2) followed by bromoethane gives compound D, C13H20. Draw possible structures for A, B, C, and D. H₂, Lindlar's Problem 13 of 14 Structure of A 1. NaNH2 Submit 2. CH3CH₂Br Structure of product Darrow_forward-Arrange the following acids in the increasing order of their solubility in water: p-ClC6H4COOH, CH3COOH, CH3(CH2)3COOH -Which one of the following will be most acidic and why? 2-Chlorobutanoic acid, 4-Chlorobutanoic acid, Butanoic acid, 3-Chlorobutanoic acid Give the products of the following reactions.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

