
Concept explainers
Interpretation:
The structure of the productformed by intramolecular Diels–-Alder reaction of the given compound at room temperature is to be drawn.
Concept introduction:
>The conjugate addition of
Dienophile is the alkene that adds to diene.Diene is an electron-rich system attacksan electron deficient dienophile.The diene and dienophile, when residing in a single molecule, the intramolecular Diels–-Alder reaction is possible.
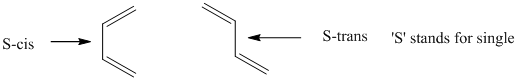
>In order to form the cyclic transition state in Diels–-Alder reaction, the conjugated diene must be able to adopt
The
Want to see the full answer?
Check out a sample textbook solution
Chapter 11 Solutions
ORGANIC CHEMISTRY (LOOSELEAF)-PACKAGE
- 2,3-Di-tert-butyl-1,3-butadiene does not undergo Diels–Alder reactions. Explain.arrow_forwardComplete the Diels-Alder reactions by drawing structures for the products in each question.arrow_forwardIdentify the starting materials that would be needed to produce the Diels-Alder products shown below.arrow_forward
- What represents the structure of the diene to carry out the following Diels-Alder reaction?arrow_forwardIntramolecular Diels–Alder reactions are possible when a substrate contains both a 1,3-diene and a dienophile, as shown in the following general reaction.With this in mind, draw the product when each compound undergoes an intramolecular Diels–Alder reaction.arrow_forwardWhat is the structure of the Diels Alder product according to this reaction?arrow_forward
- Draw the mechanism of the diels alder reaction between anthracene (diene) and the dienophile maleic anhydride. The structure of anthracene is provided belowarrow_forwardWhy does the diels alder reaction often show high selectivity and predictable stereochemistry?arrow_forwardRank the following dienes in order of increasing reactivity in a Diels–Alder reaction.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

