
Solutions Manual for Organic Chemistry
10th Edition
ISBN: 9781259636387
Author: Carey Dr., Francis A
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 11.2, Problem 3P
The two compounds shown differ by a factor of
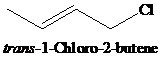
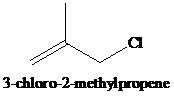
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Explain the Summary of Factors That Determine Whether the SN1 or SN2 Mechanism Occurs ?
The reaction of tert-butyl bromide with azide ion (N3-) in methanol is a typical SN1 reaction. What happens to the rate of the reaction if [N3-] is doubled?
Can you do 5,6,7 and explain it? State the mechanism in which through reaction proceeds sn2 or sn1 and can you explain the process of the mechanism
Chapter 11 Solutions
Solutions Manual for Organic Chemistry
Ch. 11.1 - Prob. 1PCh. 11.1 - Prob. 2PCh. 11.2 - The two compounds shown differ by a factor of 60...Ch. 11.2 - Prob. 4PCh. 11.2 - Prob. 5PCh. 11.2 - Prob. 6PCh. 11.3 - Prob. 7PCh. 11.3 - Prob. 8PCh. 11.3 - Evaluate 2,3,3-trimethyl-1-butene as a candidate...Ch. 11.4 - After heating a solution of allyl tert-butyl...
Ch. 11.5 - Prob. 11PCh. 11.6 - Prob. 12PCh. 11.8 - Prob. 13PCh. 11.9 - What dienes containing isolated double bonds are...Ch. 11.10 - Prob. 15PCh. 11.10 - Prob. 16PCh. 11.11 - Prob. 17PCh. 11.12 - Dicarbonyl compounds such as quinones are reactive...Ch. 11.12 - 2,3-Di-tert-butyl-1,3-butadiene is extremely...Ch. 11.12 - Methyl acrylate (H2C=CHCO2CH3) reacts with...Ch. 11.13 - Prob. 21PCh. 11.14 - What diene and dienophile could you use to prepare...Ch. 11.14 - Write equations in the synthetic direction for the...Ch. 11.16 - Prob. 24PCh. 11.16 - Prob. 25PCh. 11 - Write structural formulas for each of the...Ch. 11 - Give an acceptable IUPAC name for each of the...Ch. 11 - A certain species of grasshopper secretes an...Ch. 11 - Which of the following are chiral?...Ch. 11 - Describe the molecular geometry expected for...Ch. 11 - Prob. 31PCh. 11 - What compound of molecular formula C6H10 gives...Ch. 11 - Prob. 33PCh. 11 - Prob. 34PCh. 11 - Prob. 35PCh. 11 - Prob. 36PCh. 11 - Identify the more reactive dienophile in each of...Ch. 11 - Prob. 38PCh. 11 - Prob. 39PCh. 11 - Prob. 40PCh. 11 - Prob. 41PCh. 11 - Prob. 42PCh. 11 - Prob. 43PCh. 11 - Prob. 44PCh. 11 - Prob. 45PCh. 11 - Prob. 46PCh. 11 - Show how to prepare each of the following...Ch. 11 - Prob. 48PCh. 11 - Prob. 49PCh. 11 - Prob. 50PCh. 11 - Compound A was converted to compound B by the...Ch. 11 - Suggest reasonable explanations for each of the...Ch. 11 - Prob. 53PCh. 11 - Prob. 54PCh. 11 - Prob. 55DSPCh. 11 - Prob. 56DSPCh. 11 - Prob. 57DSPCh. 11 - Prob. 58DSPCh. 11 - Prob. 59DSP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Which of the species below best depicts the likely transition state for the reaction between methyl iodide, CH3I and sodium methylthiolate, NaSCH3?arrow_forwardShow the mechanism of the reaction of drawing 1 mole of water from the 2,2,5-trimethyl-3-hexanol compound, indicating the reaction conditions, step by step. Indicate the main product and by-product. b) Does the main product show the geometric isomer? If it does not show the isomers, please indicate why it does not. c) Write the products formed when the. main product ozonlamp is reduced.arrow_forwardGive 3 examples of a reaction mechanism of E1 that follows Zaitsev's rule.arrow_forward
- Does increasing the energy barrier for an SN2 reaction increase or decrease the magnitude of the rate constant for the reactionarrow_forwardThe rate law for the reaction of HO- with tert-butyl bromide to form an elimination product in 75% ethanol/ 25% water at 30 °C is the sum of the rate laws for the E2 and E1 reactions. What percentage of the reaction takes place by an E2 pathway when [HO-] = 5.0 M? (k2 = 7.1 x 10-5, and k1 = 1.5 x 10-5)arrow_forwarda. Compound 2 (see image) is required to be synthesized from two possible starting materials, which route is the best? Justify the choice through the use of structures, please show the arrows. b. Draw the mechanism for the solvolysis reaction of t-butyl bromide in isopropanol. The dielectric constant of isopropanol is 18, will the solvolysis reaction in ethanol of this substrate be slower or faster compared to isopropanol? Justify your answer. Include the arrows. c. Complete the following reaction (see image), showing each of the products that are formed. What kind of reactions are they? Justify the reason for the second step.arrow_forward
- 19. Which of the following represents the transition state of the rate-determining step in the SN1 reaction between tert-butyl chloride and water? (see attached screenshot) A. 1 B. 2 C. 3 D. 4arrow_forwardDialkyl peroxides are a family of compounds which can be explosive when heated. These explosions begin with the following bond fragmentation (which is then followed by other reaction steps). Which process best describes this fragmentation? E2 Elimination Heterolytic dissociation Homolytic dissociation Racemizationarrow_forwardPredict the products and mechanisms of the following reactions. When if more than one product or mechanism is possible, explain are the most probable. (d) 2-chloro-2-methylbutane by heating in ethanol (e) isobutyl iodide, KOH in ethanol / waterarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
Enzymes - Effect of cofactors on enzyme; Author: Tutorials Point (India) Ltd;https://www.youtube.com/watch?v=AkAbIwxyUs4;License: Standard YouTube License, CC-BY
Enzyme Catalysis Part-I; Author: NPTEL-NOC IITM;https://www.youtube.com/watch?v=aZE740JWZuQ;License: Standard Youtube License