
Concept explainers
(a)
Interpretation:
The major product should be given, when given compound is treated with acid.
Concept introduction:
Dehydration reaction:
Removal of water molecule from the reaction when the alcohol is treated with strong acid like sulfuric acid is known as dehydration reaction, for example
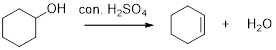
(b)
Interpretation:
The major product should be given, when given compound is treated with acid.
Concept introduction:
Dehydration reaction:
Removal of water molecule from the reaction when the alcohol is treated with strong acid like sulfuric acid is known as dehydration reaction, for example
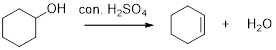
(c)
Interpretation:
The major product should be given, when given compound is treated with acid.
Concept introduction:
Dehydration reaction:
Removal of water molecule from the reaction when the alcohol is treated with strong acid like sulfuric acid is known as dehydration reaction, for example
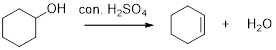
(d)
Interpretation:
The major product should be given, when given compound is treated with acid.
Concept introduction:
Dehydration reaction:
Removal of water molecule from the reaction when the alcohol is treated with strong acid like sulfuric acid is known as dehydration reaction, for example
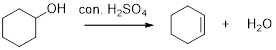
(e)
Interpretation:
The major product should be given, when given compound is treated with acid.
Concept introduction:
Dehydration reaction:
Dehydration reaction:
Removal of water molecule from the reaction when the alcohol is treated with strong acid like sulfuric acid is known as dehydration reaction, for example
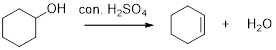
(f)
Interpretation:
The major product should be given, when given compound is treated with acid.
Concept introduction:
Dehydration reaction:
Removal of water molecule from the reaction when the alcohol is treated with strong acid like sulfuric acid is known as dehydration reaction, for example
Want to see the full answer?
Check out a sample textbook solution
Chapter 11 Solutions
Organic Chemistry-With Access and Study Guide / Solutions Manual
- What are the major products obtained when each of the following ethers is heated with one equivalent of HI?arrow_forwardDraw the products obtained when each of the following ethers is heated with one equivalent of HBr:arrow_forwardAcetal product formation is difficult, since hemiacetals are stable while acetals are not stable. True Falsearrow_forward
- Draw the product formed when each compound is treated with two equivalents of CH3CH2CH2CH2MgBr followed by H2O.arrow_forwardWhat product is formed when each compound is treated with either LiAlH4 (followed by H2O), or NaBH4 in CH3OH?arrow_forwardList three different sets of reagents (each set consisting of a carbonyl compound and a Grignard reagent) that could be used to prepare each of thefollowing tertiary alcohols:arrow_forward
- 1. Answer the following questions A.How many alcohol reagents are used to form the final product during the hemiacetal formation? B. How many reactive sites in this picture when reacted with Potassium permanganate with presence of H2SO4? Choices: 1,2,3,4arrow_forwardRank the alcohols in order of increasing reactivity when dehydrated with H2SO4.arrow_forwardDraw the organic products formed when attached allylic alcohol A is treated with following reagent. H2 + Pd-Carrow_forward
- Explain Addition of Alcohols—Acetal Formation ?arrow_forwardDraw the product formed when pentanal (CH3CH2CH2CH2CHO) is treatedwith following reagent. With some reagents, no reaction occurs. [1] C6H5Li; [2] H2Oarrow_forwardThe alcohol shown in part A was synthesised via a Grignard reaction. There are three different combinations of the Grignard reagent and carbonyl that can make the product. Draw those three Grignard reagents and carbonyls in the designated boxes.arrow_forward
 EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
