
(a)
Interpretation:
The stereo isomeric product should be given, when it is reacts with per oxyacid followed by hydroxide ion.
Concept introduction:
Formation of
The
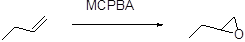
In the nucleophilic substitution reaction, the
In
Reactant and nucleophile are present at the rate determination step.
The order of species involving in
Tertiary < Secondary < Primary
(b)
Interpretation:
The stereo isomeric product should be given, when it is reacts with per oxyacid followed by hydroxide ion.
Formation of epoxide:
The alkene can be converted to epoxide, when it is treated with MCPBA (m-chloro perbenzoic acid)
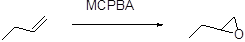
In the nucleophilic substitution reaction, the rate of reaction depends on reactant as well as nucleophile, which are involved in reaction is called bimolecular nucleophilic substitution reaction.
In
Reactant and nucleophile are present at the rate determination step.
The order of species involving in
Tertiary < Secondary < Primary
(c)
Interpretation:
The stereo isomeric product should be given, when it is reacts with per oxyacid followed by hydroxide ion.
Formation of epoxide:
The alkene can be converted to epoxide, when it is treated with MCPBA (m-chloro perbenzoic acid)
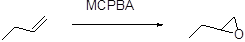
In the nucleophilic substitution reaction, the rate of reaction depends on reactant as well as nucleophile, which are involved in reaction is called bimolecular nucleophilic substitution reaction.
In
Reactant and nucleophile are present at the rate determination step.
The order of species involving in
Tertiary < Secondary < Primary
(d)
Interpretation:
The stereo isomeric product should be given, when it is reacts with per oxyacid followed by hydroxide ion.
Formation of epoxide:
The alkene can be converted to epoxide, when it is treated with MCPBA (m-chloro perbenzoic acid)
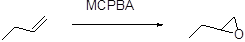
In the nucleophilic substitution reaction, the rate of reaction depends on reactant as well as nucleophile, which are involved in reaction is called bimolecular nucleophilic substitution reaction.
In
Reactant and nucleophile are present at the rate determination step.
The order of species involving in
Tertiary < Secondary < Primary
Want to see the full answer?
Check out a sample textbook solution
Chapter 11 Solutions
Organic Chemistry-With Access and Study Guide / Solutions Manual
- 8. R-MgX and CO2 are reacted to form isobutyric acid. What is R? Use IUPAC nomenclature of the substituent. 9. From which cycloalkene can heptanoic acid be formed from through oxidative cleavage? Use IUPAC name. 10. The catalytic hydrogenation of a benzene will result to this cycloalkanearrow_forwardExplain why the addition of HBr to alkenes A and C is regioselective, forming addition products B and D, respectively.arrow_forwardCHEMISTRY AND ENVIRONMENTAL POLLUTION Explain how Hg2+ ion is transformed into methylmercury anddimethylmercury in the aquatic environment.arrow_forward
- The addition reaction of an acid (HBr) to an alkene (CH3CH=CH2) follows Markovnikov's rule and involves: A) initial attack by Br– B) initial attack by Br• C) isomerization of CH3CH2CH2Br D) formation of a primary carbocation. E) formation of a secondary carbonation. (F) Formation of allyl carbocationarrow_forward2. In the bromination reactions, what is the function of CCl4? Why can it fulfil its role?3. Bromination proceeds by either free radical substitution or electrophillic addition. Based on Table 3, which mechanism is followed by alkanes? by alkenes? by alkynes?4. For which hydrocarbon type is light necessary for bromination to take place?5. What is the function of light in the bromination reaction? Why are alkenes and alkynes not included as samples?arrow_forwarddraw the two possible carbocations that can form when this alkene reacts with a strong acid (such as HBr or H3O+). of the two structures you drew, circle the more stable carbocationarrow_forward
- What is the major product of the reaction between 2-methylbut-2-ene and Br2 in H2O?arrow_forwardDraw the product or products that will be obtained from the reaction of cis-2-butene and trans-2-butene with each of the following reagents. If a product can exist as stereoisomers, show which stereoisomers are formed. 1. HCl. 4.Br2 in CH2Cl2 7. H2O + H2SO4 2. BH3/THF, followed by HO-, H2O2, H2O 5. Br2 + H2O 8. CH3OH + H2SO4 3. a peroxyacid 6. H2 + Pd/Carrow_forwardExplain Stability of Alkenes ?arrow_forward
- Explain the 2 step addition of bromide to an alkene.arrow_forwardThe same alkane is obtained from the catalytic hydrogenation of both alkene A and alkene B. The heat of hydrogenation of alkene A is 29.8 kcal/mol, and the heat of hydrogenation of alkene B is 31.4 kcal/mol. Which alkene is more stable?arrow_forwardWhat two stereoisomeric alkanes are formed from the catalytic hydrogenation of (z)-3-methyl-2-hexene? Draw and label the alkanes. What are the relative amounts of each alkane?arrow_forward

