
ORGANIC CHEMISTRY
4th Edition
ISBN: 9781119745105
Author: Klein
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Question
Chapter 11.4, Problem 8ATS
Interpretation Introduction
Interpretation: An efficient synthesis for the following transformation is to be determined.
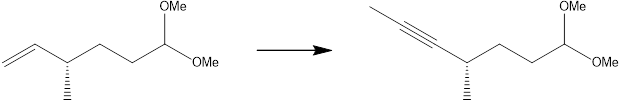
Concept introduction: One carbon atom must be added to the final product in this process. Because the carbon-carbon triple bond in the final product has taken the place of the carbon-carbon double bond in the starting material, there is also a change in the identity of a
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
In the mid-1930s a substance was isolated from a fungus that is a parasite of ryes and other grasses. This alkaloid, lysergic acid, has
been of great interest to chemists because of its strange, dramatic action on the human mind. Many derivatives of lysergic acid are
known, some with medicinal applications. Perhaps the best known derivative of lysergic acid is the potent hallucinogen lysergic acid
diethylamide (LSD):
మగవా జి
N-H
LSD
(CH25N;O)
Like other alkaloids, LSD is a weak base, with Kp = 7.6 × 107. What is the pH of a 0.94 M solution of LSD?
pH =
Following are structural formulas for amphetamine and methamphetamine.
H
NH,
CH3
(a)
(b)
Amphetamine
(racemic)
Methamphetamine
(racemic)
The major central nervous system effects of amphetamine and amphetamine-like
drugs are locomotor stimulation, euphoria and excitement, stereotyped behavior, and
anorexia. Show how each drug can be synthesized by reductive amination of an ap-
propriate aldehyde or ketone and amine.
Arrange the members of each group in order of decreasing basicity: (a) Ammonia, aniline, methylamine (b) Acetanilide, aniline, N-methylaniline (c) 2,4-Dichloroaniline, 2,4-dimethylaniline, 2,4-dinitroaniline (d) 3,4-Dichloroaniline, 4-chloro-2-nitroaniline, 4-chloro-3-nitroaniline (e) Dimethylamine, diphenylamine, N-methylaniline
Chapter 11 Solutions
ORGANIC CHEMISTRY
Ch. 11.1 - Prob. 1CCCh. 11.1 - Prob. 2CCCh. 11.2 - Prob. 1LTSCh. 11.2 - Prob. 3PTSCh. 11.3 - Prob. 2LTSCh. 11.3 - Prob. 6ATSCh. 11.4 - Prob. 3LTSCh. 11.4 - Prob. 7PTSCh. 11.4 - Prob. 8ATSCh. 11.5 - Prob. 4LTS
Ch. 11.5 - Propose an efficient synthesis for each of the...Ch. 11.5 - Prob. 10ATSCh. 11 - Prob. 11PPCh. 11 - Prob. 12PPCh. 11 - Prob. 13PPCh. 11 - Prob. 14PPCh. 11 - Prob. 15PPCh. 11 - Prob. 16PPCh. 11 - Prob. 17PPCh. 11 - Prob. 18PPCh. 11 - Prob. 19PPCh. 11 - Prob. 20PPCh. 11 - Prob. 21PPCh. 11 - Prob. 22PPCh. 11 - Prob. 23PPCh. 11 - Prob. 24PPCh. 11 - Prob. 25PPCh. 11 - Prob. 26ASPCh. 11 - Prob. 27ASPCh. 11 - Prob. 28ASPCh. 11 - Prob. 29ASPCh. 11 - Prob. 30ASPCh. 11 - Prob. 31ASPCh. 11 - Prob. 32ASPCh. 11 - Prob. 33IPCh. 11 - Prob. 34IPCh. 11 - Prob. 35IPCh. 11 - Prob. 36IPCh. 11 - Prob. 37IPCh. 11 - Prob. 38IPCh. 11 - Prob. 39IPCh. 11 - Prob. 42CPCh. 11 - The compound Z-3-hexenyl acetate is one of several...Ch. 11 - When a consumer purchases a tomato, smell is one...Ch. 11 - Prob. 45CPCh. 11 - Prob. 46CP
Knowledge Booster
Similar questions
- Paroxetine (Paxil) is an antidepressant that is a member of a family of drugs known as Selective Serotonin Reuptake Inhibitors (SSRIS). This family of drugs also includes fluoxetine (Prozac) and sertraline (Zoloft). SSRIS work by inhibiting the reuptake of the neurotransmitter serotonin in the synapses of the central nervous system follow- ing release of serotonin during excitation of individual nerve cells. Between firings, the serotonin is taken back up by a nerve cell in preparation for firing again. Inhibition of reuptake has the effect of increasing the time serotonin molecules remain in the syn- apses following excitation, leading to a therapeutic effect. In one synthesis of parox- etine, the following reagents are used. Draw the structures of synthetic intermediates A and B. F НО SOCI, A B HO Pyridinearrow_forwardGiven this retrosynthetic analysis, propose a synthesis for bupropion.arrow_forwardPhenacetin is an analgesic compound having molecular formula C10H13NO2. Once a common component in over-the-counter pain relievers such as APC (aspirin, phenacetin, caffeine), phenacetin is no longer used because of its liver toxicity. Deduce the structure of phenacetin from its 1H NMR and IR spectra.arrow_forward
- Compound A undergoes an acid-catalyzed hydrolysis. One of the products (B) that is isolated gives the following 1H NMR spectrum. Identify the compounds A and Carrow_forwardShow how to prepare the following aromatic amines by aromatic nitration, followed by reduction. You may use benzene andtoluene as your aromatic starting materials.(a) anilinarrow_forward18arrow_forward
- Quinapril (trade name Accupril) is used to treat high blood pressure and congestive heart failure. One step in the synthesis of quinapril involves reaction of the racemic alkyl bromide A with a single enantiomer of the amino ester B. (a) What two products are formed in this reaction? (b) Given the structure of quinapril, which one of these two products is needed tosynthesize the drug?arrow_forwardFollowing is a synthesis for toremifene, a nonsteroidal estrogen antagonist whose structure is closely related to that of tamoxifen. (a) This synthesis makes use of two blocking groups, the benzyl (Bn) group and the tetrahydropyranyl (THP) group. Draw a structural formula of each group and describe the experimental conditions under which it is attached and removed. (b) Discuss the chemical logic behind the use of each blocking group in this synthesis. (c) Propose a mechanism for the conversion of D to E. (d) Propose a mechanism for the conversion of F to toremifene. (e) Is toremifene chiral? If so, which of the possible stereoisomers are formed in this synthesis?arrow_forwardAs far back as the 16th century, South American Incas chewed the leaves of the coca bush, Erythroxylon coca, to combat fatigue. Chemical studies of Erythroxylon coca by Friedrich Wöhler in 1862 resulted in the discovery of cocaine, C17H21NO4, as the active component. Basic hydrolysis of cocaine leads to methanol, benzoic acid, and another compound called ecgonine, C9H15NO3. Oxidation of ecgonine with CrO3 yields a keto acid that readily loses CO2 on heating, giving tropinone. (a) What is a likely structure for the keto acid? (b) What is a likely structure for ecgonine, neglecting stereochemistry? (c) What is a likely structure for cocaine, neglecting stereochemistry?arrow_forward
- Phenacetin, a substance formerly used in over-the-counter headache remedies, has the formula C10H13NO2. Phenacetin is neutral and does not dissolve in either acid or base. When warmed with aqueous NaOH, phenacetin yields an amine, C8H11NO, whose 1H NMR spectrum is shown. When heated with HI, the amine is cleaved to an aminophenol, C6H7NO. What is the structure of phenacetin, and what are the structures of the amine and the aminophenol?arrow_forwardThe reaction of cyclohexanone and N,N-dimethylamine with sodiumtriacetoxyborohydride (NaBH(OAc)3) in methanol with acetic acid (AcOH) affords N,Ndimethylcyclohexanamine in high yield. However, if anhydrous HCl is used the reaction yield of N,Ndimethylcyclohexanamine is low and a side reaction occurs to produce alcohol. Draw a reaction mechanism for the reductive amination sequence, highlighting tautomers present in the reaction.arrow_forwardSeveral sulfonylureas, a class of compounds containing RSO2NHCONHR, are useful drugs as orally active replacements for injected insulin in patients with adult-onset diabetes. These drugs decrease blood glucose concentrations by stimulating b cells of the pancreas to release insulin and by increasing the sensitivity of insulin receptors in peripheral tissues to insulin stimulation. Tolbutamide is synthesized by the reaction of the sodium salt of p-toluenesulfonamide and ethyl N-butylcarbamate . Propose a mechanism for this step.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning


Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning