
(a)
Interpretation:
The compound which undergoes metathesis to form the following compound has to be identified.
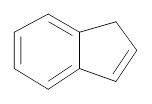
Concept introduction:
Alkene Metathesis Or (olefin metathesis)
This breaks the double bond of an alkene and then rejoins the fragments. When the fragments are joined, the new double bond is formed between two
Terminal alkene gives the best yields of a single alkene product in metathesis because one of the products is ethane, which is equally removed from the reaction mixture, thus shifting the equilibrium in favor of the other new alkene product.
(b)
Interpretation:
The compound which undergoes metathesis to form the following compound has to be identified.
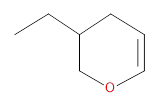
Concept introduction:
Alkene Metathesis Or (olefin metathesis)
This breaks the double bond of an alkene and then rejoins the fragments. When the fragments are joined, the new double bond is formed between two
Terminal alkene gives the best yields of a single alkene product in metathesis because one of the products is ethane, which is equally removed from the reaction mixture, thus shifting the equilibrium in favor of the other new alkene product.
Want to see the full answer?
Check out a sample textbook solution
Chapter 11 Solutions
EBK ORGANIC CHEMISTRY
- complete the following synthesesarrow_forwardDraw the main organic product formed in each of the following reactions:arrow_forwardOne step in the synthesis of occidentalol, a natural product isolated from the eastern white cedar tree, involves the following reaction. Identify the structure of A and show how A is converted to B.arrow_forward
- Identify and name the product in each of the following reactions.arrow_forwardWhat are the major organic products obtained from the following reaction?arrow_forwardBy taking into account electronegativity differences, draw the products formed by heterolysis of the carbon–heteroatom bond in each molecule. Classify the organic reactive intermediate as a carbocation or a carbanion.arrow_forward
- Complete the synthesis of the following compound from the starting material givenarrow_forwardSynthesize the product from the given material. Give the reagents necessary and draw out any intermediate products along the way.arrow_forwardExplain why the reaction in Problem 18.48 proceeds dramatically more slowly under neutral conditions than under either acidic or basic conditions.arrow_forward
