
(a)
Interpretation:
The three dimensional structure of one unit cell of the alkali metal fulleride superconductor
Concept introduction:
Unit cell is defined as the smaller group of atoms having overall crystal symmetry from which one can build the whole lattice in repetition order in 3D form.
(b)
Interpretation:
The number of
Concept introduction:
The shape of the tetrahedral holes is triangular. When 2 tetrahedral holes combine from different layers, an octahedral hole is formed. Thus, due to alignment of tetrahedral holes of first and second order align together, an octahedral hole is formed.
(c)
Interpretation:
The fractional coordinates needs to be determined for all the octahedral and tetrahedral holes in the unit cell.
Concept introduction:
Fractional coordinates are defined as fractions of edge length of the unit cell. For example, at the center of the cell, the fractional coordinate of a hole is
(d)
Interpretation:
The radii of the octahedral and tetrahedral holes needs to be calculated assuming the
Concept introduction:
The face diagonal in the unit cell can be represented as follows:
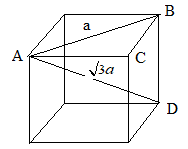
The length of a face diagonal is
Now, along the body diagonal there are 2 spheres touching the tetrahedral holes of radius r thus,
Here, rt is radius of the tetrahedral hole.
Or,
On rearranging,
Here, R is radius of the sphere.
Similarly, an octahedral hole can be represented as follows:
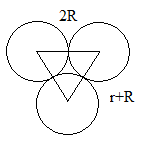
Here, radius of sphere is R and that of an octahedral hole is
Or,
Or,
Here, R is radius of the sphere.
(e)
Interpretation:
The ion from the given ions that will fit into the octahedral and tetrahedral holes needs to be determined. The ion that will only fit if the framework of
Concept introduction:
The shape of the tetrahedral holes is triangular. When 2 tetrahedral holes combine from different layers, an octahedral hole is formed. Thus, due to alignment of tetrahedral holes of first and second order align together, an octahedral hole is formed.
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
CHEMISTRY-MOD.MASTERING (18W)
- A unit cell of Re03 consists of a cubic .1 arrangement of Re atoms with O atoms centred along each edge of the cube. The coordination number of each Re atom isarrow_forwardBy making use of CFT in tetrahedral and octahedral geometries, predict whether nickel ferrite mineral (NiFe2O4) has a normal spinel or inverse spinel structure. Explain the structure of this mineral in detail.arrow_forwardWhat are the applications of superconductors?arrow_forward
- Discuss the need for a high-Tc superconductor.arrow_forward1. Tantalum is rare, hard, blue-gray, lustrous transition metal that is highly corrosion-resistant. It has a density of 16.4 kg/L and a molecular weight of 180.948 g/mol. What is the (A) atomic radius in cm, (B) volume in cm^3 of Ta if it adopts the body centered cubic unit structure and (C) volume of unit cellarrow_forward(a) What change occurs when AgCl is doped with CdCl2?(b) What type of semiconductor is produced when silicon is doped with boron?arrow_forward
- Give information about the Nickel Arsenide structure (NiAs). For example: What kind of ionic solid is it, what are its coordination properties, and what is the cation/anion radius ratio? Give information.arrow_forwardBased on its phase diagram, explain the formation of cored structure during the solidification of Ni-Cu alloy.arrow_forwardHow many lattice atoms surround a tetrahedral hole and an octahedral hole in a closest-packed structure? Describe which hole site is larger and explain why.arrow_forward
- In a metallurgical process the mineral pyrite, FeS2, is roasted in air: FeS 2 +O 2 Fe 2 O 3 +SO 2 The SO 2 is then converted into H 2 SO 4 in the following reactions: 2SO 2 +O 2 2SO 3; SO 3 +H 2 SO 4 H 2 S 2 O 7; H 2 S 2 O 7 +H 2 O 2H 2 SO 4 Assuming the mineral is 24.0\%FeS 2 and the remainder is inert, what mass of H 2 SO 4 is produced if 155 g of the mineral is used?arrow_forward(a) Determine the number of calcium ions in the chemicalformula of the mineral hardystonite, CaxZn(Si2O7). (b)Determine the number of hydroxide ions in the chemicalformula of the mineral pyrophyllite, Al2(Si2O5)2(OH)x.arrow_forwardWhat colour are SnC12.2H2O crystals?arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning



