
Interpretation:
The compound(s) that are more soluble in water has to be predicted from the following options –
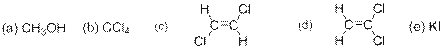
Concept Introduction:
Solubility of a substance depends on various factors like – chemical structure of the substance, temperature, pH etc,
Like dissolves like is a Universal rule which states that polar compounds dissolve more in polar solvents and non-polar compounds dissolve more in non-polar compounds.
Dipole moment is the measure of polarity in polar covalent compounds.
Answer to Problem 12.1KSP
Options (a), (d) and (e) are correct answers.
Explanation of Solution
Reason for correct answers:
The compound given in the options (a), (d) and (c) are polar compounds and so they dissolve in water which is a polar solvent.
The compound given in option (a) is methanol. It is a polar covalent compound as the
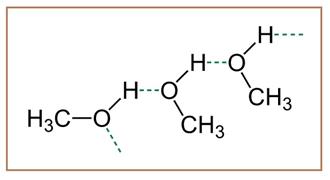
Figure 1
The compound given in option (d) is cis-1,2-dichloroethene. The
The compound given in option (e) is Potassium iodide and it is an ionic compound made of
Reasons for Incorrect answers:
The compounds given in options (
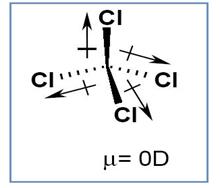
Figure 2
Though the
Therefore polarity of the compound gets cancelled.
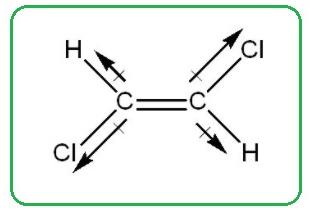
Figure 3
The compound(s) that are more soluble in water has been predicted from the given options.
Want to see more full solutions like this?
Chapter 12 Solutions
Chemistry: Atoms First
- Cooing Curve of Water i) What phase(s) at each of the numbered sections?ii) At what temperature is this substance a) condensing and b) freezing?iii) At which number section(s) is/are kinetic energy of the molecule is the greatest?iv) Relate the answer at (iii) to the associated intermolecular force of the molecules.arrow_forwardThe lowest land on earth is located along the shoreline of the Dead Sea at approximately 430 m below sea level. At this altitude, air pressure is 1.05 atm. At what temperature should water boil here? (I think you can use Clausius-Clapeyron, but I'm not sure.)arrow_forwardwhich if the following is true about hydrogen bonding?arrow_forward
- Differentiate between the surface tension of water and that of alcohol. You’ve seen small insects walk on water. Would they be able to walk on alcohol? Explain your answer. No plagarism please!arrow_forwardConsider the following molecules: Na2O, HCl, NH3, N2, CH3CH2OH. Which would have the highest viscosity? please follow below steps to answer the question. Indicate a 'plan' on how you intend to solve YOUR problem. You should have a numerical list. These can be phrases (not complete sentences) if you want, as long as the material is sufficiently easy to follow. Steps should be detailed. For example, "look in the book for the equation" does not work as you could not use that step while taking exams. Show the step by step solution of the problem. Using your plan, (and labeling each step), solve your predicted exam question. Show your work. For example, if you have a scientific notation question, you CANNOT say "move the decimal" but must illustrate each step of the process. If one step requires dimensional analysis to solve you can show your set up using arrows like I do in our videos - but show all of your work. Explain each step of your process. Arrive at the answer in…arrow_forwardThe temperature inside a pressure cooker is 115 C. Calculate the vapor pressure of water inside the pressure coker. What would be the temperature inside the pressure cooker if the vapor pressure of water was 3.50 atm? Can you plz explain using a simple manner, Im horrible at math also use the clausius i provided below.arrow_forward
- What is the chemical reason (ie. molecular property) for biodiesel having more desirable viscosity and solidification temperature properties than vegetable oil?arrow_forward2. A compound boils at 125oC and has a ∆Hvapof 45 kJ/mol. What is the compound's vapor pressure at 18oC? Answer to 3 significant figures and in mmHg.arrow_forwardThe hydrogen fluoride molecule, HF, is more polar than a water molecule, H2O (for example, has a greater dipole moment), yet the molar enthalpy of vaporization for liquid hydrogen fluoride is lesser than that for water.Explain.arrow_forward
- Is molecules located on the surface of a liquid lack neighbors in the surface tenison? True or Falsearrow_forwardAs the intermolecular attractive forces between moleculesincrease in magnitude, do you expect each of the followingto increase or decrease in magnitude? (a) Vapor pressure,(b) heat of vaporization, (c) boiling point, (d) freezing point,(e) viscosity, (f) surface tension, (g) critical temperature.arrow_forwardThe vapor pressure of ethanol is 400. mmHg at 63.5°C. Its molar heat of vaporization is 39.3 kJ/mol. What is vapor pressure of ethanol, in mmHg, at (3.90x10^1)°C? Note: Your answer is assumed to be reduced to the highest power possible. Answer: ______x10___ _____unitsarrow_forward
 World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
