
(a)
Interpretation:
How the given compound can be produced from an alkene is to be shown.
Concept introduction:
Electrophilic addition of carbenes to
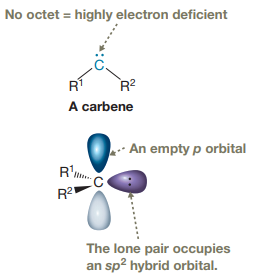
Carbenes are typically highly electrophilic due to the lack of an octet and are
Answer to Problem 12.2P
The given compound can be produced from an alkene is shown below:
Explanation of Solution
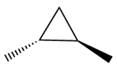
The given compound is:
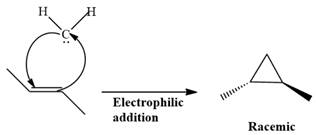
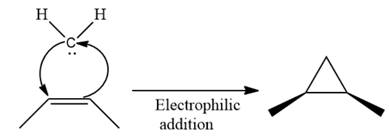
Electrophilic addition of carbenes to alkenes forms cyclopropane rings. Diazomethane (
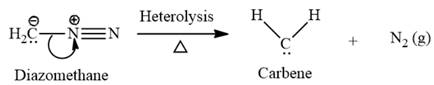
This carbene intermediate reacts with an alkene in a concerted one-step reaction to form a cyclopropyl ring. The bridge carbon atom in the cyclopropyl ring comes from the carbene while the two carbon atoms are from the original alkene. Two new
![]()
The complete reaction of the addition of carbene to alkene is shown below:
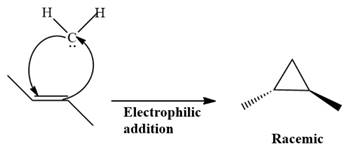
The given compound is produced via an electrophilic addition reaction of a carbene to an alkene.
(b)
Interpretation:
How the given compound can be produced from an alkene is to be shown.
Concept introduction:
Electrophilic addition of carbenes to alkenes forms cyclopropane rings. In carbine, the carbon atom has two bonds and a lone pair of electrons.

Carbenes are typically highly electrophilic due to the lack of an octet and are
Answer to Problem 12.2P
The given compound can be produced from an alkene is shown below:

Explanation of Solution
The given compound is:
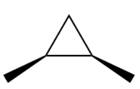
Electrophilic addition of carbenes to alkenes forms cyclopropane rings. Diazomethane (
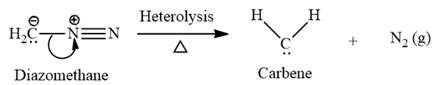
This carbene intermediate reacts with an alkene in a concerted one-step reaction to form a cyclopropyl ring. The bridge carbon atom in the cyclopropyl ring comes from the carbene while the two carbon atoms are from the original alkene. Two new
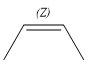
The complete reaction of the addition of carbene to alkene is shown below:
The given compound is produced via an electrophilic addition reaction of a carbene to an alkene.
(c)
Interpretation:
How the given compound can be produced from an alkene is to be shown.
Concept introduction:
Electrophilic addition of carbenes to alkenes form cyclopropane rings. In carbine, the carbon atom has two bonds and a lone pair of electrons.

Carbenes are typically highly electrophilic due to the lack of an octet and are
Answer to Problem 12.2P
The given compound can be produced from an alkene is shown below:

Explanation of Solution
The given compound is:
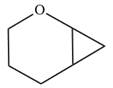
Electrophilic addition of carbenes to alkenes forms cyclopropane rings. Diazomethane (
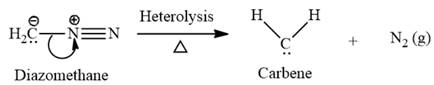
This carbene intermediate reacts with an alkene in a concerted one-step reaction to form a cyclopropyl ring. The bridge carbon atom in the cyclopropyl ring comes from the carbene while the two carbon atoms are from the original alkene. Two new
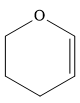
The complete reaction of the addition of carbene to alkene is shown below:
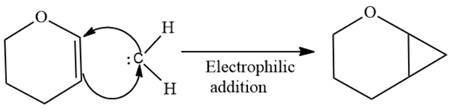
The given compound is produced via an electrophilic addition reaction of a carbene to an alkene.
(d)
Interpretation:
How the given compound can be produced from an alkene is to be shown.
Concept introduction:
Electrophilic addition of carbenes to alkenes forms cyclopropane rings. In carbine, the carbon atom has two bonds and a lone pair of electrons.

Carbenes are typically highly electrophilic due to the lack of an octet and are
Answer to Problem 12.2P
The given compound can be produced from an alkene is shown below:
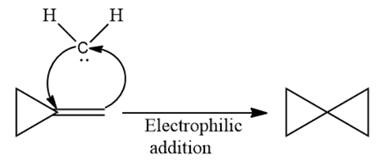
Explanation of Solution
The given compound is:

Electrophilic addition of carbenes to alkenes forms cyclopropane rings. Diazomethane (
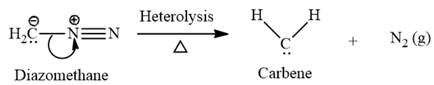
This carbene intermediate reacts with an alkene in a concerted one-step reaction to form a cyclopropyl ring. The bridge carbon atom in the cyclopropyl ring comes from the carbene while the two carbon atoms are from the original alkene. Two new
The complete reaction of the addition of carbene to alkene is shown below:
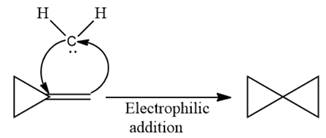
The given compound is produced via an electrophilic addition reaction of a carbene to an alkene.
Want to see more full solutions like this?
Chapter 12 Solutions
ORGANIC CHEMISTRY:PRINCIPLES...(CL)
- This is a hydrogenation and carbene addition problem. How do I find what the missing reactant is of the reaction?arrow_forwardPredict the major product of each of the reactions shown here and provide the complete, detailed mechanism.arrow_forward(SYN) Show how to produce each of the following compounds from a hydrocarbon that has the formula indicatedarrow_forward
- Illustrate details mechanism of reactions for bromination of the following alkane. State each of the steps involves.arrow_forwardPredict/draw the major product of the reaction shown in the picture:arrow_forwardPlease answer question below, be very deatiled and draw out the mechanism reaction with arrowsarrow_forward
- Draw the complete, detailed mechanism for the reaction shown here. Will the product be optically active? Explain.arrow_forwardDraw the complete, detailed mechanism for each of the following reactions and predict the major products.arrow_forwarddraw mechanism for below reaction, name the final productarrow_forward
- (SYN) Show how you would synthesize each of these compounds from benzene.arrow_forwardBy following the curved red arrows, draw the product(s) of each of the following reaction steps. Also indicate which species is the electrophile and which is the nucleophile.arrow_forwardWhich of the following addition reactions is not likely to occur? Why? (Please explain clearly, have already done problem)arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





