
Concept explainers
Stearidonic acid (C18H28O2) is an unsaturated fatty acid obtained from oils isolated from hemp and blackcurrant
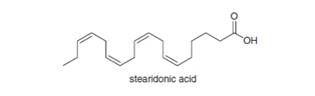
a. What fatty acid is formed when stearidonic acid is hydrogenated with excess
b. What fatty acids are formed when stearidonic acid is hydrogenated with one equivalent of
c. Draw the structure of a possible product formed when stearidonic acid is hydrogenated with one equivalent of
d. How do the melting points of the following fatty acids compare: stearidonic acid; one of the products formed in part (b); the product drawn in part (c)?
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
ORGANIC CHEMISTRY SOLUTION MANUAL
Additional Science Textbook Solutions
Thermodynamics, Statistical Thermodynamics, & Kinetics
Introduction to Chemistry
Chemistry: The Central Science (13th Edition)
Chemistry
General Chemistry: Principles and Modern Applications (11th Edition)
Organic Chemistry (9th Edition)
- Between fatty acid A and B, Which is most likely solid at room temperature? CH3(CH2)18CO2H CH3CH2CH=CHCH2CH=CHCH2CH=CH(CH2)7CO2Harrow_forward1. Some marine plankton contain triacylglycerols formed from the polyunsaturated fatty acid (PUFA) such as: CH3CH2CH=CHCH2CH=CHCH2CH=CHCH2CH=CHCH2CH=CHCH2COOH a) Draw the skeletal structure of the given PUFA showing the preffered stereogenic arrangement for a naturally-occuring lipid at each double bond. b) Write the systematic name of the fatty acid. c) What type of omega-n acid is this fatty acid? d) Would you expect this fatty acid to be a soild or liquid at room temperature?arrow_forwardConsider erucic acid whose formula is CH3(CH2)7CH=CH(CH2)11COOH. It isa.Two of the choices are correctb.an omega-9 saturated fatty acidc.an omega-9 fatty acidd.an omega-7 unsaturated fatty acide.an unsaturated fatty acidarrow_forward
- Which fatty acid has the lowest melting point? Question 2 options: CH3(CH2)10COOH CH3(CH2)16COOH CH3(CH2)14COOH CH3(CH2)12COOHarrow_forward1) The starch in 25kg (~1 bushel) of corn is hydrolyzed and in the process 1.8kg of water (H2O) is consumed. How many moles of glucose (C6H12O6) would you expect to be produced? C6H12O6 -> 2 CO2 + 2 C2H5OH. 2) If the glucose from the above hydrolysis is fermented, how many moles of ethanol (C2H5OH) would result? How many grams? 3) If ethanol has a density of 0.789g/ml, what would the volume of the ethanol be in ml? What would the volume be in gallons (1gal = 3785ml)? 4) The above ethanol could be converted to Vodka by diluting to 50% ethanol (adding enough water to double the volume). What would the volume of the resulting Vodka be in ml? If the volume of a “shot” of Vodka is 30ml, how many shots of vodka could be produced from a bushel of corn?arrow_forwardWhich of the following is an omega 6 fatty acid? Fatty acid A: CH3(CH2)5CH=CH(CH2)7COOH Fatty acid B: CH3(CH2)8COOH Fatty acid C: CH3(CH2)7CH=CH(CH2)7COOH Fatty acid D: CH3(CH2)4CH=CH(CH2)7COOHarrow_forward
- By saponification of plant material and subsequent acidification, petroselenic acid (C18:1Δ6), which was forming a triglyceride, is extracted. Mark the INCORRECT option: a) The petroselenic acid was part of a vegetable oil. b) Petroselenic acid is classified as ω6. c) Petroselenic acid has a double bond between C6 and C7. d) The oil from which the petroselenic acid was extracted is liquid due to its unsaturation. e) The saponification of said oil produces a salt of petroselenic acid (soap).arrow_forwarda) Draw structural formulas for the two monosaccharides that result when structure III istreated as shown below. (b) Draw the monosaccharide that results when product A from the previous step (Q1a) istreated with Br2/H2O. (c) Draw the monosaccharide that results when product A from the previous step (Q1a) istreated with 1.NaBH4 / 2. H2O.arrow_forwardGentiobose is found in saffron. Gentribose contains two glucose molecules linked by a beta (1 - 6) glycosidic bond. Describe how to draw the glycosidic linkage in this structurearrow_forward
- Draw the structure of cyclic adenosine monophosphate (cAMP), a messenger involved in the regulation of glucose production in the body. Cyclic AMP has a phosphate ring connecting the 39- and 59-hydroxyl groups on adenosine.arrow_forward1 Hyaluronic acid acts as a lubricant in the synovial fluid of joints. In rheumatoid arthritis, inflammation breaks hyaluronic acid down to smaller molecules. Under these conditions, what happens to the lubricating power of the synovial fluid?arrow_forwardWrite equations for the following reactions, representing the reactants and products using structural formulas. (a) The formation of ethyl acetate from acetic acid and ethanol (b) The hydrolysis of glyceryl tristearate (the triester of glycerol with stearic acid, a fatty acid; Table 23.9)arrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning





