
Answer the following questions about alcohol B.
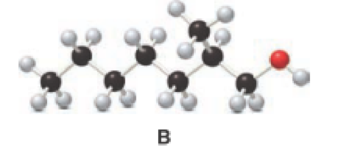
- a. Give the IUPAC name.
- b. Classify the alcohol as 1°, 2°, or 3°.
- c. Draw the products formed when B is dehydrated with H2SO4.
- d. What product is formed when B is oxidized with K2Cr2O7?
- e. Draw a constitutional isomer of B that contains an OH group.
- f. Draw a constitutional isomer of B that contains an ether.
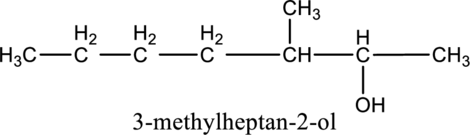
a.
Interpretation:
IUPAC name for a given alcohol compound has to be determined.
Concept introduction:
Nomenclature of alcohol:
Firstly, find the longest carbon chain, which is bonded with the
Explanation of Solution
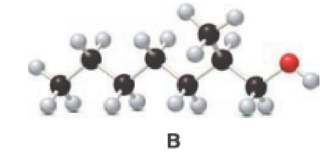
Given compound B is,
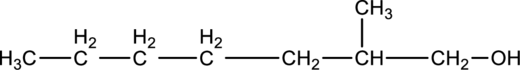
Step 1: Step 1: In a given compound the black, white and red color balls indicates carbon, hydrogen and oxygen atoms. In the given compound the longest carbon chain, which is attached to the
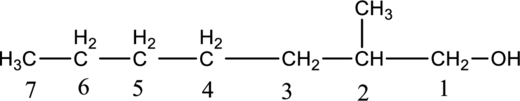
Step 2: For naming the compound, firstly, the parent carbon chain should be checked, and as it possess seven carbon atoms, it is named as heptane. Due to
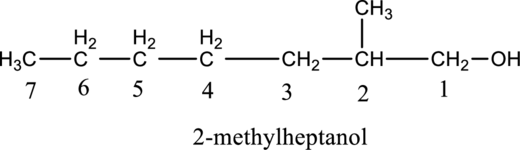
b.
Interpretation:
From the compound B, the type of alcohol has to be determined.
Concept introduction:
Classification of alcohol:
Generally, the alcohol group is bonded with minimum one alkyl group. The alcohol group is classified into three types, such as primary
Explanation of Solution
In the below compound A,
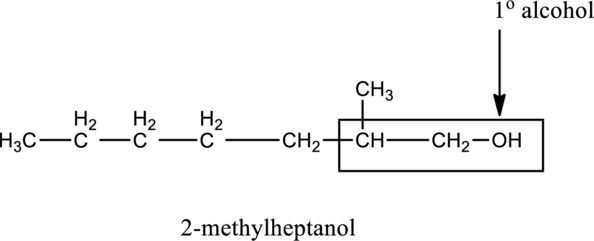
c.
Interpretation:
The product obtained when the compound B reacts with
Concept introduction:
Dehydration of alcohol:
When alcohol reacts with
Explanation of Solution
Given stating material B is 2-methylheptanol
When 2-methylheptanol reacts with
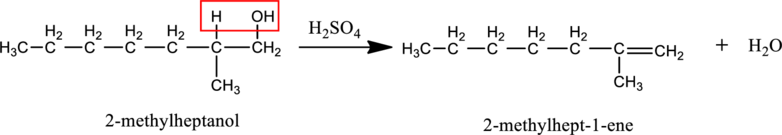
d.
Interpretation:
The product obtained when the compound B reacts with K2Cr2O7 reagent has to be determined.
Concept introduction:
Oxidation of alcohol:
During oxidation of alcohol, the number of
Explanation of Solution
Given stating material A is 2-methylheptanol
When 2-methylheptanol, the primary alcohol containing two hydrogen atom on the carbon attached with
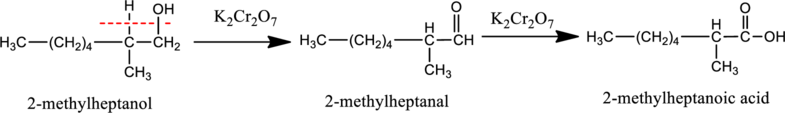
e.
Interpretation:
The constitutional isomer of compound B that contain
Concept introduction:
Constitutional isomers:
The compounds, that contain same molecular formula but different with respect to molecular orientation. In other words compounds having similar molecular formula and different structure are called as Constitutional isomers.
Explanation of Solution
In the given compound B molecular formula is
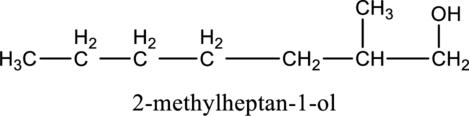
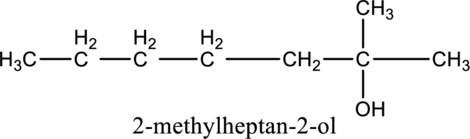
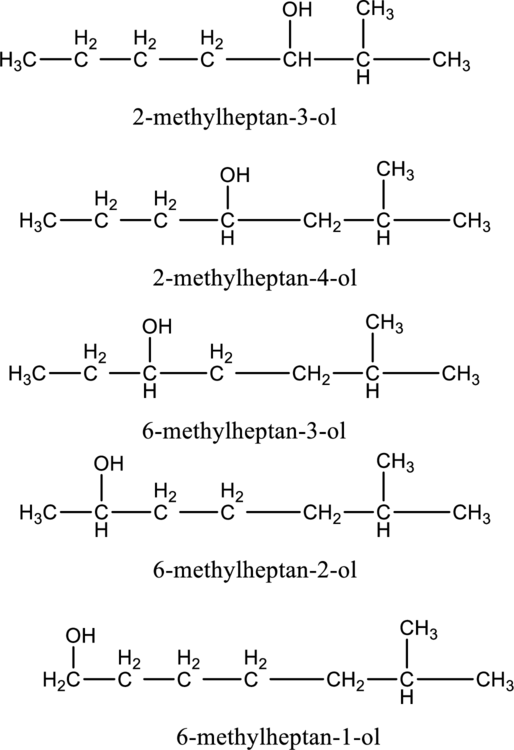
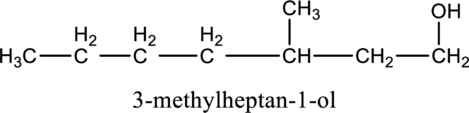
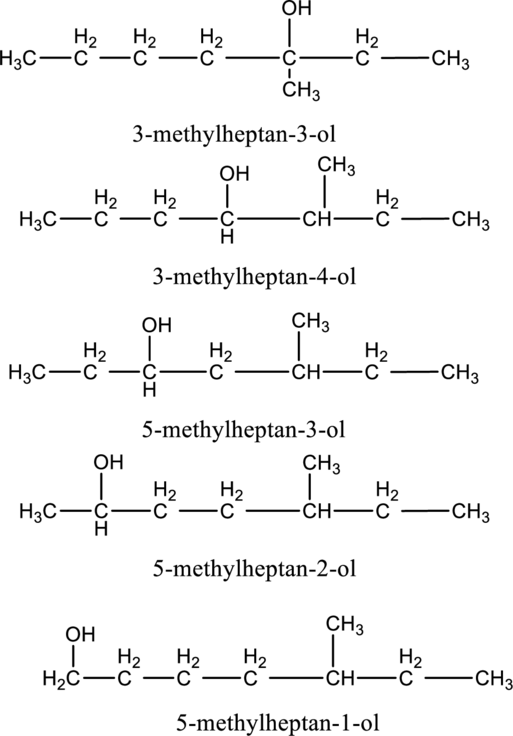
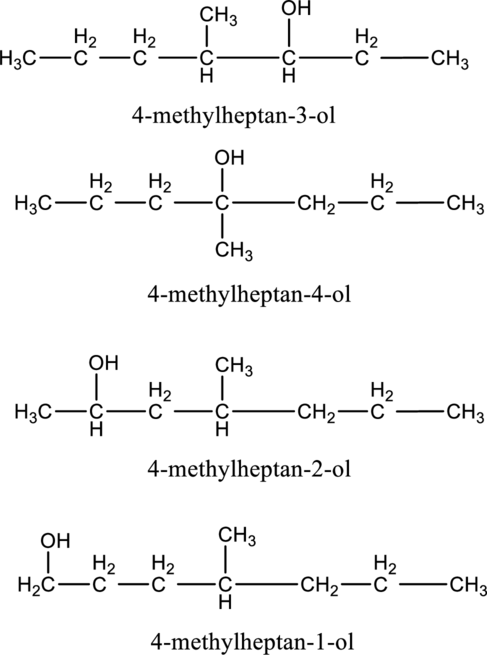
f.
Interpretation:
The constitutional isomer of compound B that contain an ether group has to be determined.
Concept introduction:
Refer to part ‘e.’.
Explanation of Solution
In the given compound A molecular formula is
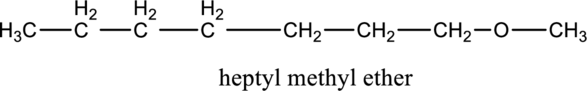
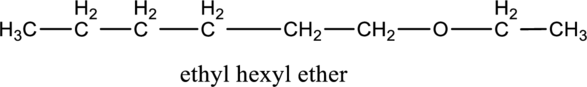
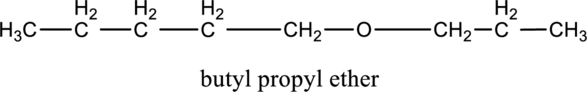
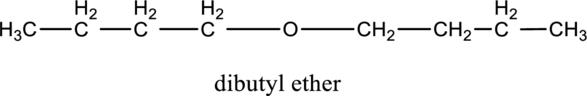
Want to see more full solutions like this?
Chapter 12 Solutions
General, Organic, & Biological Chemistry
- n-Butyl methyl ether is an isomer of MTBE and has a boiling point of 70 oC. Explain why the boiling point is significantly different compared to MTBE.arrow_forwardAnswer each question using the ball-and-stick model of compound A.a.Give the IUPAC name for A, including R,S designations for stereogenic centers. b.Classify A as a 1°, 2°, or 3° alcohol. c.Draw a stereoisomer for A and give its IUPAC name. d.Draw a constitutional isomer that contains an OH group and give its IUPAC name. e.Draw a constitutional isomer that contains an ether and give its IUPAC name. f.Draw the products formed (including stereochemistry) when A is treated with each reagent: [1] NaH; [2] H2SO4; [3] POCl3, pyridine; [4] HCl; [5] SOCl2, pyridine; [6] TsCl, pyridinearrow_forwardIdentify the IUPAC name of the given structure. A. 2 - methylhexan-5-one B. 5 - methylhexan-2-one C. 2 - heptanone D. 5 - heptanone Identify the IUPAC name of the given structure. A. 4 - bromopentan-3-one B. 1 - bromobutan-2-one C. 2 - bromobutan-one D. None of the abovearrow_forward
- Draw the product resulting from mild oxidation of (a) 2-butanol; (b) 2-methylpropanal; (c) cyclopentanol.arrow_forwardMolecule Type Boiling point (°C) CH3CH2CH3 Alkane -42 CH3CHO Aldehyde +21 CH3CH2OH Alcohol +78 i. Why is the boiling point of the aldehyde greater than that of the alkane?ii. Why is the boiling point of alcohol the highest?iii. Explain why the solubility of aldehydes and alcohols falls as the molecules get bigger.arrow_forwardExplain the method to prepare an ether ?arrow_forward
- Glycerol contains: a. oxygens which are each bonded to two alkyl groups b. oxygens single-bonded to primary and secondary carbons c. Oxygens double-bonded to carbon, with alkyls on both sides d. Oxygens double-bonded to carbon, with alkyls on one side only e. Oxygens double-bonded to carbon, with an alkyl on one side and an --OH on the other sidearrow_forwardgive the structure corresponding to each IUPAC name 1. 6,6-diethyl-4-nonanolarrow_forwardGive the IUPAC name for each alcohol.arrow_forward
- Give three reasons why ethers make good laboratory reagentsarrow_forward1.Write the balanced equations for all the organic substances in the lucas test.( Oraganic substances: ethyl alcohol,isopropyl alcohol, t-butyl alcohol and resorcinol)2. What is the purpose of adding acetone in the oxidation of alcohols? 3. What simple test can be used to distinguish alcohols from phenols?arrow_forwardCategorize the alcohol belowarrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Introductory Chemistry: A FoundationChemistryISBN:9781285199030Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781285199030Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning


