
(a)
Interpretation:
The detailed mechanism with the major product for the given reaction is to be drawn.
Concept introduction:
The acid-catalyzed hydration of an
Answer to Problem 12.42P
The detailed mechanism for the given reaction is
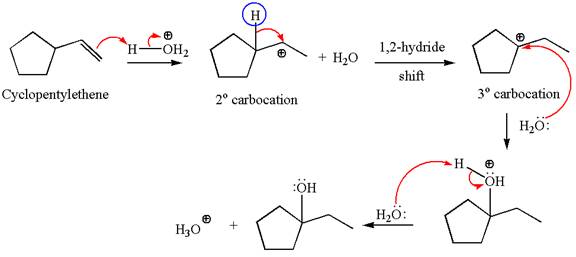
Explanation of Solution
The given reaction is
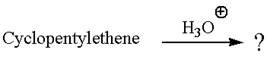
Cyclopentylethene, a terminal alkene, in presence of
The first step is the formation of a secondary carbocation by proton transfer reaction. The proton transfers to the less substituted double bonded carbon.
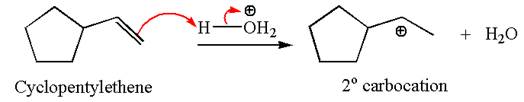
The secondary carbocation can be rearranged to a more stable tertiary carbocation by
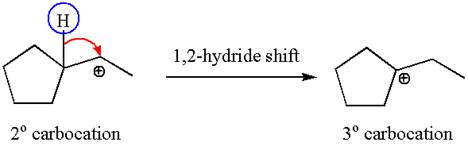
In the second step, the water molecule acts as a nucleophile and attacks the tertiary carbocation, forming a tertiary alcohol product, followed by deprotonation of the positively charged oxygen.
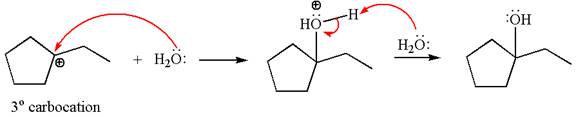
The detailed mechanism for the given reaction is drawn by suggesting that the reaction occurs through carbocation rearrangement.
(b)
Interpretation:
The detailed mechanism with major product for the given reaction is to be drawn.
Concept introduction:
The oxymercuration-reduction is also the reaction of addition of water across the
Answer to Problem 12.42P
The detailed mechanism for the given reaction is
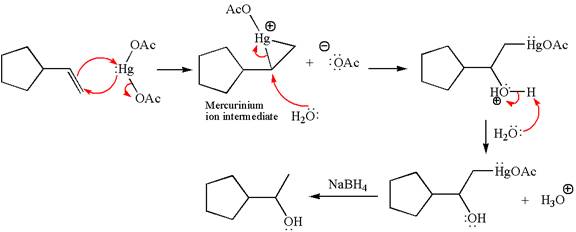
Explanation of Solution
The given reaction is
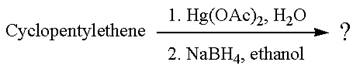
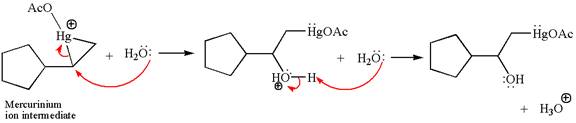
Cyclopentylethene, a terminal alkene, on reaction with mercury
In the first step, the electron rich
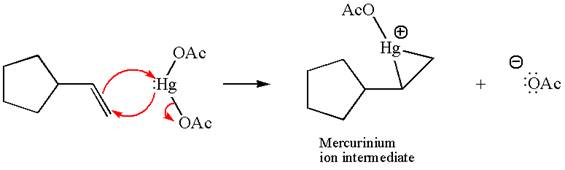
In the second step, the water molecule acts as a nucleophile and, according to Markovnikov rule, attacks the most substituted side to open the three-membered ring, followed by deprotonation of the positively charged oxygen atom.
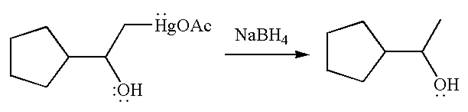
The product formed in the previous step is then subjected to reduction with sodium borohydride,
The detailed mechanism for the given reaction is drawn by suggesting that the reaction occurred through formation of mercurinium ion intermediate without rearrangement.
(c)
Interpretation:
The detailed mechanism with major product for the given reaction is to be drawn.
Concept introduction:
The acid-catalyzed hydration of an alkene is the electrophilic addition of water across the
Answer to Problem 12.42P
The detailed mechanism for the given reaction is
Explanation of Solution
The given reaction is
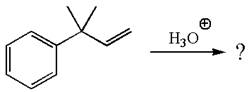
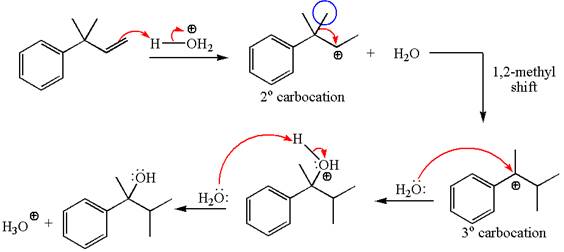
The given substrate, a terminal alkene, in the presence of
The first step is the formation of a secondary carbocation by proton transfer reaction. The proton transfers to the less substituted double bonded carbon.
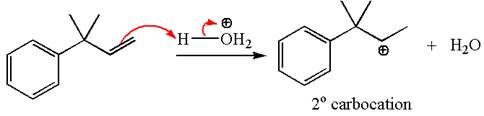
The secondary carbocation can be rearranged to a more stable tertiary as well as resonance stabilized carbocation by
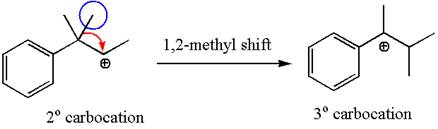
In the second step, the water molecule acts as a nucleophile and attacks the tertiary carbocation, forming a tertiary alcohol product, followed by deprotonation of the positively charged oxygen.
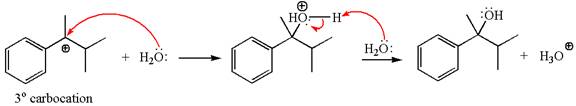
The detailed mechanism for the given reaction is drawn by suggesting that the reaction occurred through carbocation rearrangement.
(d)
Interpretation:
The detailed mechanism with major product for the given reaction is to be drawn.
Concept introduction:
The oxymercuration-reduction is also the reaction of addition of water across the
Answer to Problem 12.42P
The detailed mechanism for the given reaction is
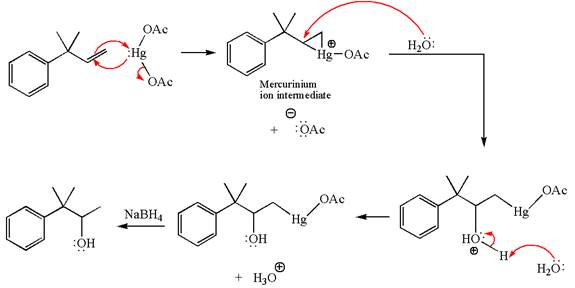
Explanation of Solution
The given reaction is
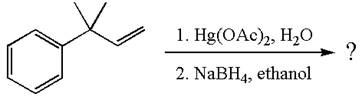
The given substrate, a terminal alkene, on reaction with mercury
In the first step, the electron rich
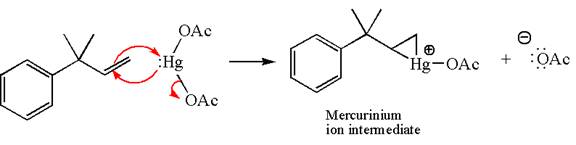
In the second step, the water molecule acts as a nucleophile and, according to Markovnikov rule, attacks the most substituted side to open the three-membered ring, followed by deprotonation of the positively charged oxygen atom.
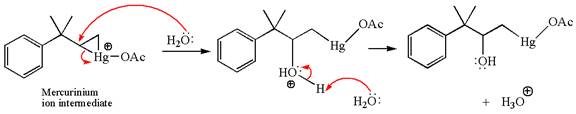
The product formed in the previous step is then subjected to reduction with sodium borohydride,
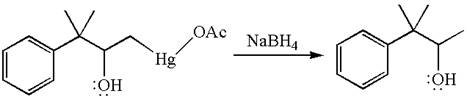
The detailed mechanism for the given reaction is drawn by suggesting that the reaction occurred through formation of mercurinium ion intermediate without rearrangement.
Want to see more full solutions like this?
Chapter 12 Solutions
ORGANIC CHEMISTRY PRINCIPLES & MECHANISM
- Draw the complete, detailed mechanism for the reaction shown here and give the major product. CH3I (excess) ? NH2arrow_forwardOH Draw the complete, detailed mechanism for the reaction shown here. (See Problem 17.63.) NaBH4 Ethanolarrow_forwardDraw the complete, detailed mechanism for the following reaction.arrow_forward
- Draw a complete, detailed mechanism for the following reaction. A key intermediate is provided.arrow_forwardDraw the complete, detailed mechanism for the following reaction along with the major product. Cl2 HOẠCarrow_forwardDraw the complete, detailed mechanism for each of the following reactions ( (b) NaOH ? U NaOH ?arrow_forward
- Draw the complete, detailed mechanism for the following reaction.arrow_forwardShown below is a two-step mechanism beginning with nucleophilic attack of water, and subsequent deprotonation with a base. Draw the arrows for the mechanism for both step 1 and step 2 and draw the intermediate product of in the box. + H-O OH + H₂Oarrow_forwardDraw the complete, detailed mechanism for each of the following reactions and predict the major product(s). (a) (b) Br,/HO ? Br,/H H2O H2Oarrow_forward
- Draw the complete mechanism of each pair of reactants including any favorable rearrangements and all important resonance structures of all intermediates. a. Which reaction has a lower PE carbocation intermediate? b. Draw an energy diagram showing the reaction profiles of both reactions in the previous question. Use a dotted line for the first pair of reactants and a solid line for the second pair of reactants. (Assume the energy of the starting materials and products are the same for both pairs and the reactions are neither uphill nor downhill on net. c. Mark points on the energy diagram corresponding to each carbocation in your mechanisms.arrow_forwardDraw a complete, detailed mechanism for the reaction shown here.arrow_forwardDraw the mechanism and both products for the following reaction. Lil THFarrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
