
Loose Leaf for Chemistry: The Molecular Nature of Matter and Change
8th Edition
ISBN: 9781260151749
Author: Silberberg Dr., Martin; Amateis Professor, Patricia
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Question
Chapter 12, Problem 12.76P
Interpretation Introduction
Interpretation:
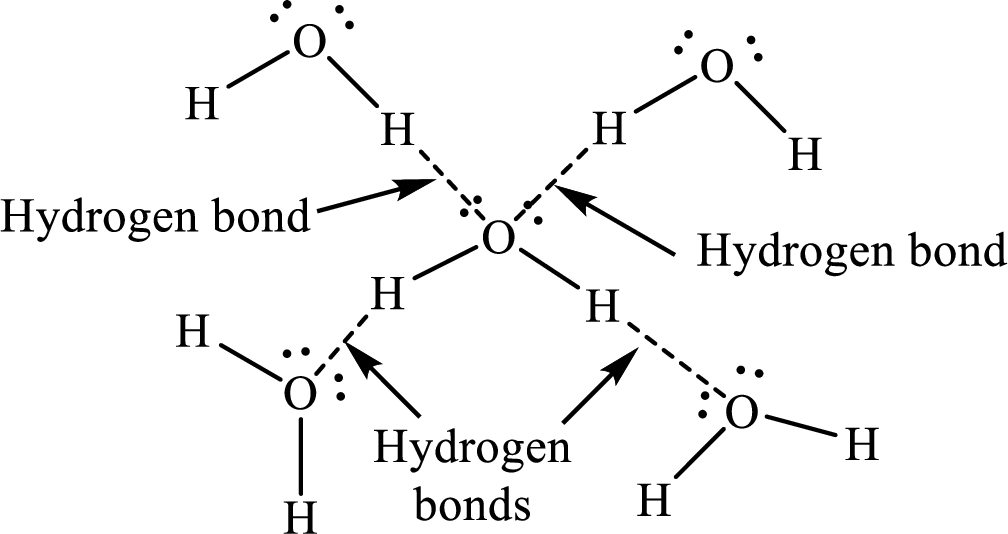
The molecular terms that occur when the ice melts are to be described.
Concept introduction:
Water
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
Loose Leaf for Chemistry: The Molecular Nature of Matter and Change
Ch. 12.2 - Prob. 12.1AFPCh. 12.2 - Prob. 12.1BFPCh. 12.2 - Prob. 12.2AFPCh. 12.2 - Prob. 12.2BFPCh. 12.2 - Prob. 12.3AFPCh. 12.2 - Prob. 12.3BFPCh. 12.3 - Prob. 12.4AFPCh. 12.3 - Prob. 12.4BFPCh. 12.3 - Prob. 12.5AFPCh. 12.3 - Prob. 12.5BFP
Ch. 12.6 - For each of the following crystalline solids,...Ch. 12.6 - Prob. 12.6BFPCh. 12.6 - Prob. 12.7AFPCh. 12.6 - Iron crystallizes in a body-centered cubic...Ch. 12.6 - Prob. 12.8AFPCh. 12.6 - Prob. 12.8BFPCh. 12.6 - Prob. B12.1PCh. 12.6 - Prob. B12.2PCh. 12 - Prob. 12.1PCh. 12 - Prob. 12.2PCh. 12 - Prob. 12.3PCh. 12 - Prob. 12.4PCh. 12 - Prob. 12.5PCh. 12 - Prob. 12.6PCh. 12 - Prob. 12.7PCh. 12 - Name the phase change in each of these events: (a)...Ch. 12 - Prob. 12.9PCh. 12 - Many heat-sensitive and oxygen-sensitive solids,...Ch. 12 - Prob. 12.11PCh. 12 - Prob. 12.12PCh. 12 - Prob. 12.13PCh. 12 - Prob. 12.14PCh. 12 - Prob. 12.15PCh. 12 - Prob. 12.16PCh. 12 - Prob. 12.17PCh. 12 - Prob. 12.18PCh. 12 - From the data below, calculate the total heat (in...Ch. 12 - Prob. 12.20PCh. 12 - Prob. 12.21PCh. 12 - Prob. 12.22PCh. 12 - Prob. 12.23PCh. 12 - Prob. 12.24PCh. 12 - Prob. 12.25PCh. 12 - Prob. 12.26PCh. 12 - Prob. 12.27PCh. 12 - Prob. 12.28PCh. 12 - Prob. 12.29PCh. 12 - Prob. 12.30PCh. 12 - Use Figure 12.10 to answer the following:
Carbon...Ch. 12 - Prob. 12.32PCh. 12 - Prob. 12.33PCh. 12 - Prob. 12.34PCh. 12 - Prob. 12.35PCh. 12 - Prob. 12.36PCh. 12 - Distinguish between polarizability and polarity....Ch. 12 - Prob. 12.38PCh. 12 - Prob. 12.39PCh. 12 - Prob. 12.40PCh. 12 - Prob. 12.41PCh. 12 - Prob. 12.42PCh. 12 - Prob. 12.43PCh. 12 - Prob. 12.44PCh. 12 - Prob. 12.45PCh. 12 - Prob. 12.46PCh. 12 - Prob. 12.47PCh. 12 - Prob. 12.48PCh. 12 - Prob. 12.49PCh. 12 - Which liquid in each pair has the lower vapor...Ch. 12 - Which substance has the lower boiling point?...Ch. 12 - Which substance has the higher boiling point?...Ch. 12 - Prob. 12.53PCh. 12 - Prob. 12.54PCh. 12 - Prob. 12.55PCh. 12 - Prob. 12.56PCh. 12 - Why does the antifreeze ingredient ethylene glycol...Ch. 12 - Prob. 12.58PCh. 12 - Prob. 12.59PCh. 12 - Why does an aqueous solution of ethanol (CH3CH2OH)...Ch. 12 - Prob. 12.61PCh. 12 - Prob. 12.62PCh. 12 - Prob. 12.63PCh. 12 - Prob. 12.64PCh. 12 - Prob. 12.65PCh. 12 - Prob. 12.66PCh. 12 - Prob. 12.67PCh. 12 - Prob. 12.68PCh. 12 - Prob. 12.69PCh. 12 - Prob. 12.70PCh. 12 - Prob. 12.71PCh. 12 - Prob. 12.72PCh. 12 - Prob. 12.73PCh. 12 - Prob. 12.74PCh. 12 - Prob. 12.75PCh. 12 - Prob. 12.76PCh. 12 - Prob. 12.77PCh. 12 - Prob. 12.78PCh. 12 - Prob. 12.79PCh. 12 - Prob. 12.80PCh. 12 - Prob. 12.81PCh. 12 - Prob. 12.82PCh. 12 - Prob. 12.83PCh. 12 - Prob. 12.84PCh. 12 - Besides the type of unit cell, what information is...Ch. 12 - What type of unit cell does each metal use in its...Ch. 12 - What is the number of atoms per unit cell for each...Ch. 12 - Calcium crystallizes in a cubic closest packed...Ch. 12 - Chromium adopts the body-centered cubic unit cell...Ch. 12 - Prob. 12.90PCh. 12 - Prob. 12.91PCh. 12 - Prob. 12.92PCh. 12 - Prob. 12.93PCh. 12 - Prob. 12.94PCh. 12 - Prob. 12.95PCh. 12 - Prob. 12.96PCh. 12 - Prob. 12.97PCh. 12 - Prob. 12.98PCh. 12 - Prob. 12.99PCh. 12 - Prob. 12.100PCh. 12 - Prob. 12.101PCh. 12 - Prob. 12.102PCh. 12 - Prob. 12.103PCh. 12 - Polonium, the Period 6 member of Group 6A(16), is...Ch. 12 - Prob. 12.105PCh. 12 - Prob. 12.106PCh. 12 - Prob. 12.107PCh. 12 - Prob. 12.108PCh. 12 - Prob. 12.109PCh. 12 - Prob. 12.110PCh. 12 - Prob. 12.111PCh. 12 - Prob. 12.112PCh. 12 - Prob. 12.113PCh. 12 - Prob. 12.114PCh. 12 - Prob. 12.115PCh. 12 - Prob. 12.116PCh. 12 - Prob. 12.117PCh. 12 - Prob. 12.118PCh. 12 - Prob. 12.119PCh. 12 - Prob. 12.120PCh. 12 - Prob. 12.121PCh. 12 - Prob. 12.122PCh. 12 - Prob. 12.123PCh. 12 - Prob. 12.124PCh. 12 - Prob. 12.125PCh. 12 - Prob. 12.126PCh. 12 - Bismuth is used to calibrate instruments employed...Ch. 12 - Prob. 12.128PCh. 12 - Prob. 12.129PCh. 12 - Prob. 12.130PCh. 12 - Prob. 12.131PCh. 12 - Prob. 12.132PCh. 12 - Prob. 12.133PCh. 12 - Prob. 12.134PCh. 12 - Prob. 12.135PCh. 12 - Prob. 12.136PCh. 12 - Prob. 12.137PCh. 12 - Prob. 12.138PCh. 12 - Prob. 12.139PCh. 12 - Prob. 12.140PCh. 12 - Prob. 12.141PCh. 12 - Prob. 12.142PCh. 12 - Prob. 12.143PCh. 12 - Prob. 12.144PCh. 12 - Prob. 12.145PCh. 12 - The crystal structure of sodium is based on the...Ch. 12 - Prob. 12.147PCh. 12 - One way of purifying gaseous H2 is to pass it...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
Chemical Equilibria and Reaction Quotients; Author: Professor Dave Explains;https://www.youtube.com/watch?v=1GiZzCzmO5Q;License: Standard YouTube License, CC-BY