
Concept explainers
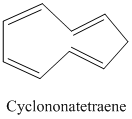
Evaluate each of the following processes applied to cyclononatetraene, and decide whether
the species formed is
Addition of one more π electron, to give
Addition of two more π electrons, to give
Loss of H+ from the
Loss of H+ from one of the
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
Loose Leaf for Organic Chemistry
- Draw all aromatic hydrocarbons that have molecular formula C8H10. For each compound, determine how many isomers of molecular formula C8H9Br would be formed if one H atom on the benzene ring were replaced by a Br atom.arrow_forwardDETERMINE EACH ITEM IF TRUE OR FALSE 1. The circle in a benzene ring represents the electrons that are moving in the entire molecule 2. A Carbon-Halogen bond is stronger as you go up the periodic table 3. C10H18 is gaseous in nature 4. Ethers can be explosive when oxygenated 5. In order for a compound to be categorized as aromatic, it must have a p orbital on every atom.arrow_forwardMeta directors in electrophilic aromatic substitution reactions have A) unpaired electrons in the atom attached to the ring B) partial or full positive charge on the atom attached to the ring C) highly electronegative atoms attached to the ring an so or sp2 hybridized atom attached to the ringarrow_forward
- Choose one answer for the following. Refer to the diagram below. 1. Hybridization of atom labelled A (sp, sp2, or sp3) 2. Hybridization of atom labelled B (sp, sp2, or sp3) 3. Hybridization of atom labelled C (sp, sp2, sp3) 4.Class compound for functional group D (phenols, aryl halide, alcohols, aromatics, or carboxylic acids) 5. Class compound for functional group E (phenols, aryl halide, alcohols, aromatics, or carboxylic acids)arrow_forwardFor the aromatic compounds below, draw the p-orbitals for the compound.arrow_forwardSelect all statements that are true regarding tertiary radicals - Tertiary radicals are more stable than primary radicals due to increased hyperconjugation from attached alkyl groups. - The CH bond that leads to a tertiary radical via homolysis has a higher bond dissociation energy that the CH bond leading to a primary radical - Tertiary radical hybridization geometry is best described as sp2 hybridization rather than sp3 hybridization - Radical chlorination reactions are more regioselective than radical bromination reactions when a tertiary radical can form.arrow_forward
- Which statement best describes the following pair of compounds? C4H6 and C6H12 A.) Circulated functional groups do not show absorption in the infrared spectrum because they have the same mass B.) The reduced mass of both compounds is the same so they do not absorb IR (infrared) energy. C.) The functional group specified in both compounds does not undergo changes in the dipole moment due to symmetry D.) Both represent an absorption band around 2000 cm ^ -1arrow_forwardMeasure the C=C-C bond angles in the energy-minimized models of the cis and trans isomers of 2,2,5,5-tetramethyl-3-hexene. In which case is the deviation from VSEPR predictions greater?arrow_forwardWhat are the overlapping orbitals between nitrogen and carbon in aniline? sp2-sp2 p-p sp3-sp3 sp3-sp2arrow_forward
- Which of the following statements about carbon radicals is not true?A) Carbon radicals are classified as primary, secondary, tertiary, or quaternary.B) A carbon radical is sp2 hybridized.C) The geometry of a carbon radical is trigonal planar.D) The unhybridized p orbital in a carbon radical contains the unpaired electron.arrow_forward1. What is resonance theory? State five conclusionstgan can be drawn from the theory. 2.What factors confer aromaticity to an organic molecule. 3. What are the various ways by which alkenes can be synthesized. 4. State the two main experiment that were used to establish the extra stability of the benzene molecule.arrow_forwardDraw structural formulas for all linear (not ring) constitutional isomers that have the molecular formula C4H4 and indicate which of the following they possess: 1. sp hybridized carbon atoms 2. sp2 hybridized carbon atoms 3. sp3 hybridized carbon atoms 4. sigma bonds, pi bondsarrow_forward

 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

