
(a)
Interpretation:
A multistep synthesis of the given compounds from the given starting material has to be given.
Concept introduction:
Bromination:

2-methyl propane undergoes radical bromination which yields the 2-bromo-2-methylpropane.because bromination will occur where the tertiary radical is present. (bromination reactions are more selective reaction).
Formation of
The
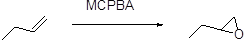
(b)
Interpretation:
A multistep synthesis of the given compounds from the given starting material has to be given.
Concept introduction:
Bromination:

2-methyl propane undergoes radical bromination which yields the 2-bromo-2-methylpropane.because bromination will occur where the tertiary radical is present. (bromination reactions are more selective reaction).
Oxidation of alcohol:
Alcohols reacts with hypochlorous (oxidizing agent) in the presence of acetic acid which yields the corresponding
Primary alcohols gives aldehyde, secondary alcohols gives ketone.
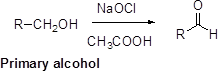
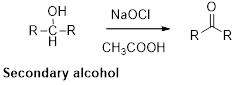
The alcohols reacts with acids like hydrochloric acid or hydrobromic acid, the bromine atom attacks back side of the carbon atoms which is bearing alcohol group and yields the corresponding product.
(c)
Interpretation:
A multistep synthesis of the given compounds from the given starting material has to be given.
Concept introduction:
Bromination:

2-methyl propane undergoes radical bromination which yields the 2-bromo-2-methylpropane.because bromination will occur where the tertiary radical is present. (bromination reactions are more selective reaction).
Oxidation of alcohol:
Alcohols reacts with hypochlorous (oxidizing agent) in the presence of acetic acid and yields the corresponding aldehyde and ketones.
Primary alcohols gives aldehyde, secondary alcohols gives ketone.
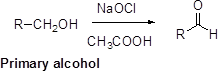
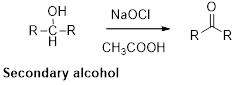
SN2 reaction:
The alcohols reacts with acids like hydrochloric acid or hydrobromic acid, the bromine atom attacks back side of the carbon atoms which is bearing alcohol group and yields the corresponding product.
(d)
Interpretation:
A multistep synthesis of the given compounds from the given starting material has to be given.
Concept introduction:
Bromination:

2-methyl propane undergoes radical bromination which yields the 2-bromo-2-methylpropane.because bromination will occur where the tertiary radical is present. (bromination reactions are more selective reaction).
Formation of epoxide:
The alkene can be converted to epoxide when alkene is treated with MCPBA (m-chloro perbenzoic acid)
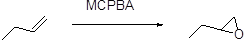
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
ORGANIC CHEMISTRY-W/S.G+SOLN.MANUAL
- Identify the best reagents to complete the following reaction. HO, CIarrow_forwardSynthesize the following compounds using cyclohexanol, or any organic reagent that has 4 or fewer carbons, and any common reagents A) но, B) онarrow_forwardIdentify the reagents to perform the following reactionarrow_forward
- Using the given starting material, any necessary inorganic reagents, and any carbon-containing compounds with no more than two carbons, indicate how the following syntheses could be carried out:arrow_forwardDevise a synthesis to prepare the product below from benzene. Complete the following reaction scheme: Part 1: Draw the structure for compound A Part 2: Draw the structure for compound B Part 3: Identify the require reagent C in the final step of the reaction. Select the single best answer.arrow_forwardDemonstrate how to convert the provided starting material into the product shown. The synthesis consists of more than one step. Show each step in the synthesisarrow_forward
- Show how the following compounds can be prepared from benzene:arrow_forwardHow can the following compounds be prepared from the given starting materials?arrow_forwardPlan a multi-step synthesis for the following starting reactant and product (electrophilic aromatic substitution emphasized): OH NO₂arrow_forward
- Ethylene oxide is the starting material for the synthesis of 1,4-dioxane. Propose a mechanism for each step in this synthesis.arrow_forwardClaisen rearrangement of an allyl phenyl ether with substituent groups in both ortho positions leads to the formation of a para-substituted product. Propose a mechanism for the following rearrangement.arrow_forwardHow can the following compounds be prepared using ethyne as the starting material?arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
