
EBK BASIC CHEMISTRY
5th Edition
ISBN: 8220101472335
Author: Timberlake
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 12.3, Problem 12.19QAP
Use the following table for problems 9.23 to 9.26:
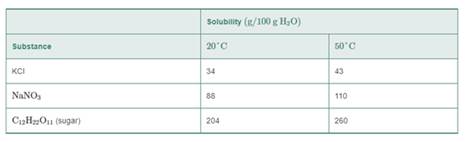
9.25 A solution containing 80 g of KCl in 200 g of H2O at 50°C is cooled at 20°C.
- How many grams of KCl remain in solution at 20°C.?
- How many grams of solid KCl crystallized after cooling?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
6. What is the boiling point of a solution that contains 190. g of MgCl₂ in 100. g of water? (Kb for
water is 0.512°C/m)
7. What is the molar mass of a compound if 5.96 g is dissolve in 25.0 g of chloroform to form a solution
which boils at 66.5°C?
{The boiling point of pure chloroform is 61.7°C & Kp = 3.63°C/m'}
a) 342 g/mol
b) 180. g/mol
c) 451 g/mol
2. A solution containing 80.g NaNO3 in 75g of H2O at 50°C is cooled to 20°C.
Solubility (g/100.g H2O)
Solubility (g/100.g H2O)
Substance
20°C
50°C
NaNO3
(a) Is the solution at 50°C saturated or unsaturated?
88
110
ch+
Chapter 12 Solutions
EBK BASIC CHEMISTRY
Ch. 12.1 - Prob. 12.1QAPCh. 12.1 - Prob. 12.2QAPCh. 12.1 - Prob. 12.3QAPCh. 12.1 - Prob. 12.4QAPCh. 12.1 - Prob. 12.5QAPCh. 12.1 - Water is a polar solvent and hexane is a nonpolar...Ch. 12.2 - Prob. 12.7QAPCh. 12.2 - Prob. 12.8QAPCh. 12.2 - Write a balanced equation for the dissociation of...Ch. 12.2 - Prob. 12.10QAP
Ch. 12.2 - Prob. 12.11QAPCh. 12.2 - Prob. 12.12QAPCh. 12.2 - Prob. 12.13QAPCh. 12.2 - Prob. 12.14QAPCh. 12.3 - Prob. 12.15QAPCh. 12.3 - State whether each of the following refers to a...Ch. 12.3 - Prob. 12.17QAPCh. 12.3 - Use the following table for problems 9.23 to 9.26:...Ch. 12.3 - Use the following table for problems 9.23 to 9.26:...Ch. 12.3 - Use the following table for problems 9.23 to 9.26:...Ch. 12.3 - Explain the following observations: More sugar...Ch. 12.3 - Explain the following observations: An open can of...Ch. 12.3 - Prob. 12.23QAPCh. 12.3 - Prob. 12.24QAPCh. 12.3 - Determine whether a solid forms when solutions...Ch. 12.3 - Prob. 12.26QAPCh. 12.4 - Prob. 12.27QAPCh. 12.4 - Prob. 12.28QAPCh. 12.4 - Calculate the mass percent (m/m) for the solute in...Ch. 12.4 - Calculate the mass percent (m/m) for the solute in...Ch. 12.4 - Calculate the mass/volume percent (m/v) for the...Ch. 12.4 - Calculate the mass/volume percent (m/v) for the...Ch. 12.4 - Prob. 12.33QAPCh. 12.4 - Calculate the grams or milliliters of solute...Ch. 12.4 - A mouthwash contains 22.5% (v/v) alcohol. If the...Ch. 12.4 - A bottle of champagne is 11% (v/v) alcohol. If...Ch. 12.4 - Prob. 12.37QAPCh. 12.4 - Prob. 12.38QAPCh. 12.4 - Prob. 12.39QAPCh. 12.4 - Prob. 12.40QAPCh. 12.4 - Prob. 12.41QAPCh. 12.4 - Prob. 12.42QAPCh. 12.4 - Prob. 12.43QAPCh. 12.4 - Prob. 12.44QAPCh. 12.4 - Prob. 12.45QAPCh. 12.4 - Prob. 12.46QAPCh. 12.4 - Prob. 12.47QAPCh. 12.4 - Prob. 12.48QAPCh. 12.4 - Prob. 12.49QAPCh. 12.4 - Prob. 12.50QAPCh. 12.5 - To make tomato soup, you add one can of water to...Ch. 12.5 - Prob. 12.52QAPCh. 12.5 - Prob. 12.53QAPCh. 12.5 - Prob. 12.54QAPCh. 12.5 - Prob. 12.55QAPCh. 12.5 - Prob. 12.56QAPCh. 12.5 - Prob. 12.57QAPCh. 12.5 - Prob. 12.58QAPCh. 12.5 - Prob. 12.59QAPCh. 12.5 - Prob. 12.60QAPCh. 12.6 - Prob. 12.61QAPCh. 12.6 - Prob. 12.62QAPCh. 12.6 - Prob. 12.63QAPCh. 12.6 - Prob. 12.64QAPCh. 12.6 - Prob. 12.65QAPCh. 12.6 - Prob. 12.66QAPCh. 12.7 - Prob. 12.67QAPCh. 12.7 - Prob. 12.68QAPCh. 12.7 - In each pair, identify the solution that will have...Ch. 12.7 - Prob. 12.70QAPCh. 12.7 - Prob. 12.71QAPCh. 12.7 - Prob. 12.72QAPCh. 12.7 - Prob. 12.73QAPCh. 12.7 - Prob. 12.74QAPCh. 12.8 - A 10% (m/v)starch solution is separated from a 1%...Ch. 12.8 - Prob. 12.76QAPCh. 12.8 - Prob. 12.77QAPCh. 12.8 - Prob. 12.78QAPCh. 12.8 - Prob. 12.79QAPCh. 12.8 - Prob. 12.80QAPCh. 12.8 - Each of the following mixtures is placed in a...Ch. 12.8 - Prob. 12.82QAPCh. 12 - Prob. 12.83FUCh. 12 - Prob. 12.84FUCh. 12 - Prob. 12.85FUCh. 12 - Prob. 12.86FUCh. 12 - Prob. 12.87UTCCh. 12 - Prob. 12.88UTCCh. 12 - Prob. 12.89UTCCh. 12 - Prob. 12.90UTCCh. 12 - Prob. 12.91UTCCh. 12 - Prob. 12.92UTCCh. 12 - Prob. 12.93UTCCh. 12 - Prob. 12.94UTCCh. 12 - Prob. 12.95UTCCh. 12 - Prob. 12.96UTCCh. 12 - Prob. 12.97AQAPCh. 12 - Prob. 12.98AQAPCh. 12 - Prob. 12.99AQAPCh. 12 - Prob. 12.100AQAPCh. 12 - Prob. 12.101AQAPCh. 12 - Prob. 12.102AQAPCh. 12 - Prob. 12.103AQAPCh. 12 - Prob. 12.104AQAPCh. 12 - If NaCl has a solubility of 36.0 g in 100. g of...Ch. 12 - Prob. 12.106AQAPCh. 12 - Prob. 12.107AQAPCh. 12 - Prob. 12.108AQAPCh. 12 - Prob. 12.109AQAPCh. 12 - Prob. 12.110AQAPCh. 12 - Prob. 12.111AQAPCh. 12 - Prob. 12.112AQAPCh. 12 - Prob. 12.113AQAPCh. 12 - What is the molarity of a solution containing 15.6...Ch. 12 - Prob. 12.115AQAPCh. 12 - Prob. 12.116AQAPCh. 12 - Prob. 12.117AQAPCh. 12 - Prob. 12.118AQAPCh. 12 - Prob. 12.119AQAPCh. 12 - Prob. 12.120AQAPCh. 12 - Prob. 12.121AQAPCh. 12 - Prob. 12.122AQAPCh. 12 - Prob. 12.123AQAPCh. 12 - Prob. 12.124AQAPCh. 12 - Prob. 12.125AQAPCh. 12 - Prob. 12.126AQAPCh. 12 - Prob. 12.127AQAPCh. 12 - Cadmium reacts with HCl to produce hydrogen gas...Ch. 12 - Prob. 12.129AQAPCh. 12 - Calculate the boiling point of each of the...Ch. 12 - Prob. 12.131AQAPCh. 12 - Prob. 12.132AQAPCh. 12 - Prob. 12.133CQCh. 12 - Write the net ionic equation to show the formation...Ch. 12 - Prob. 12.135CQCh. 12 - Prob. 12.136CQCh. 12 - Prob. 12.137CQCh. 12 - Prob. 12.138CQCh. 12 - Prob. 12.139CQCh. 12 - 9.134 A solution is prepared by dissolving 22.0 g...Ch. 12 - Prob. 12.141CQCh. 12 - Prob. 12.142CQCh. 12 - Prob. 12.143CQCh. 12 - Prob. 12.144CQCh. 12 - Prob. 12.145CQCh. 12 - Prob. 12.146CQ
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Cooking A cook prepares a solution for boiling by adding12.5 g of NaCl to a pot holding 0.750 L of water. Atwhat temperature should the solution in the pot boil?Use Table 14.5 for needed data.arrow_forwardA gaseous solute dissolves in water. The solution process has H=15 kJ. Its solubility at 22C and 6.00 atm is 0.0300 M. Would you expect the solubility to be greater or less at (a) 22C and 1 atm? (a) 18C and 6 atm? (a) 15C and 10 atm? (a) 35C and 3 atm?arrow_forward(Q68) What mass (in grams) of ethylene glycol (C2H,O2) must be added to 1,312 grams of water to lower the freezing point to -1.70 °C? (3 sf)arrow_forward
- 5.) What is the predicted value of i for NaCl? 6.) Calculate the molality of the solution (m) Use the equation: Tf (solution) – Tf (solvent) = –i Kf m The freezing point of pure water is 0.00 °C Kf for water is 1.86 °C/m 7.) Mass of solvent in gramsAdd together the masses of the liquid water and ice added to the calorimeter. Convert the volume of liquid water to mass using 1.000 g/mL as the density of water (see procedure for volume used). 8.) Mass of solvent in kilogramsUse the metric definition 1 kg = 1000 g 9.) Moles of solute (mol)Use the molality to convert kg of solvent to moles of solute.arrow_forwardColligative Properties The boiling point of chloroform (CHCl3) is 61.7C. When a nonvolatile solute is added to chloroform, its temperature increases to 83C. If 287.63 g of the solute is dissolved in 500 g of chloroform, what is the kb for chloroform? (MM solute = 98 g/mole)arrow_forward89.4 mg 27.8 mg Calcium Chloride, anhyd. 560 mg Sodium Lactate, anhyd. Water for Injection ad 100 mL 47. The pediatric infusion rate for potassium is 5 mEq/hour. If 9 mL of a 39.2% solu- tion of potassium acetate (KCH302) is di- luted to 1 L of infusion solution, calculate the proper infusion rate in mL/hr.° #8. COLYTE, a colon lavage preparation con- tains the following mixture of dry powder to prepare 4 liters of solution:arrow_forward
- What is the solubility of Cd3(PO4)2 in water? (Ksp of Cd3(PO4)2 is 2.5 x 10-3³)arrow_forwardMagnesium chloride is often used to melt ice on sidewalks. Considering that the solubility of magnesium chloride (95.21 g/mol) in water is 54.3 g per 100.0 g of water, what is the lowest temperature that you would expect to be able to melt ice with magnesium chloride? Assume ideal behavior. (Kf(water)= 1.86°C/m) A. -29.2°C B. -31.8°C C. -25.0°C D. -10.6°C E. -41.0°Carrow_forward100 90 NANO 80 70 60 CaCl 50 Pb(NO)2 40 NaCi KCI 30 20 KCIO, 10 Ce,(SO 10 20 30 40 50 60 70 80 90 100 Temperature (°C) When 20 grams of KCIO3 is dissolved in 100 grams of water at 80°C, the solution is described as O saturated supersaturated O unsaturated Solubility (g of salt in 100 g H,O) FONYarrow_forward
- 35 What mass of ethylene glycol (M.W. = 62.l g/mol) antifreeze must be added to 10.0 liters of water to produce a solution that freezes at -23.3 C degrees. Density of water is lg/ml and Kf = -l.86° C/m. Select one: a. 7.8 kg b. 8.4 g c. 6.8 kg d. 5.3 kgarrow_forwardEthylene glycol [CH2(OH)CH2(OH)]is a common automobile antifreeze. The freezing point and boiling point of a solution containing 293 g of ethylene glycol and 1085 g of water are ____and _____ respectively. (Kb and Kf for water are 0.52°C/m and 1.86°C/m, respectively) 8.09 to the power of o space end exponent C comma negative 102.3 to the power of o C negative 9.09 to the power of o space end exponent C comma 112.3 to the power of o C negative 6.09 to the power of o space end exponent C comma 108.3 to the power of o C negative 8.09 to the power of o space end exponent C comma 102.3 to the power of o Carrow_forwardWhen 20 grams of potassium chlorate, KCIO3, is dissolved in 100 grams of water at 80 °C, the solution can be correctly described as: 100 90 80 NANO, 70 Pb(NO)2 KNOS 60 CaC 50 O 40 NaCl KCI 30 20 KCIO, 10 Ce,(SO 10 20 30 40 50 60 70 80 90 100 Temperature ("C) supersaturated saturated unsaturated Solubility (g of salt in 100 g H,O) CONY 00 0 K,Cr,O,arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning  General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning
Solutions: Crash Course Chemistry #27; Author: Crash Course;https://www.youtube.com/watch?v=9h2f1Bjr0p4;License: Standard YouTube License, CC-BY