
EBK BASIC CHEMISTRY
5th Edition
ISBN: 8220101472335
Author: Timberlake
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 12.3, Problem 12.20QAP
Use the following table for problems 9.23 to 9.26:
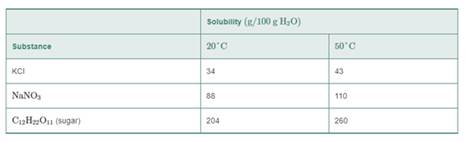
9.26 A solution containing 80 g of NaNO3 in 75 g of H2O at 50°C is cooled at 20°C.
- How many grams of NaNO3 remain in solution at 20°C.?
- How many grams of solid NaNO3 crystallized after cooling?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
At 20°C, the solubility of potassium carbonate, K2CO3, is 110.3 g/100. mL of water. In the laboratory, a
student mixes 215 g of K2CO3 with 175. g of H2O at a temperature of 20°C. (9.3)
a) How much of the K2CO3 will dissolve?
b) Is the solution saturated or unsaturated?
c) What is the mass, in grams, of any solid K2CO3 left undissolved on the bottom of the container?
The boiling point of 500.0L of water was determined to be 100.0°C. Ana has decided to increase the boiling point to
101.0°C by adding ammonium sulfate. How many grams of ammonium sulfate is required to achieve this? (Kb of H20 =
0. 512°C/m; Molar weight of ammeniam sulate=132.14g/mol)
A. 119.04 g
B. 119.12 g
C. 129.04 g
D. 129.12 g
E. None of the above.
47. A solution is made by dissolving 30.0 g of KBr in 1.00 L of water. What is the new freezing point of the
solution? The density of water is 1 g/mL. The K for water is 1.86°C/m.
(2)
f760
Chapter 12 Solutions
EBK BASIC CHEMISTRY
Ch. 12.1 - Prob. 12.1QAPCh. 12.1 - Prob. 12.2QAPCh. 12.1 - Prob. 12.3QAPCh. 12.1 - Prob. 12.4QAPCh. 12.1 - Prob. 12.5QAPCh. 12.1 - Water is a polar solvent and hexane is a nonpolar...Ch. 12.2 - Prob. 12.7QAPCh. 12.2 - Prob. 12.8QAPCh. 12.2 - Write a balanced equation for the dissociation of...Ch. 12.2 - Prob. 12.10QAP
Ch. 12.2 - Prob. 12.11QAPCh. 12.2 - Prob. 12.12QAPCh. 12.2 - Prob. 12.13QAPCh. 12.2 - Prob. 12.14QAPCh. 12.3 - Prob. 12.15QAPCh. 12.3 - State whether each of the following refers to a...Ch. 12.3 - Prob. 12.17QAPCh. 12.3 - Use the following table for problems 9.23 to 9.26:...Ch. 12.3 - Use the following table for problems 9.23 to 9.26:...Ch. 12.3 - Use the following table for problems 9.23 to 9.26:...Ch. 12.3 - Explain the following observations: More sugar...Ch. 12.3 - Explain the following observations: An open can of...Ch. 12.3 - Prob. 12.23QAPCh. 12.3 - Prob. 12.24QAPCh. 12.3 - Determine whether a solid forms when solutions...Ch. 12.3 - Prob. 12.26QAPCh. 12.4 - Prob. 12.27QAPCh. 12.4 - Prob. 12.28QAPCh. 12.4 - Calculate the mass percent (m/m) for the solute in...Ch. 12.4 - Calculate the mass percent (m/m) for the solute in...Ch. 12.4 - Calculate the mass/volume percent (m/v) for the...Ch. 12.4 - Calculate the mass/volume percent (m/v) for the...Ch. 12.4 - Prob. 12.33QAPCh. 12.4 - Calculate the grams or milliliters of solute...Ch. 12.4 - A mouthwash contains 22.5% (v/v) alcohol. If the...Ch. 12.4 - A bottle of champagne is 11% (v/v) alcohol. If...Ch. 12.4 - Prob. 12.37QAPCh. 12.4 - Prob. 12.38QAPCh. 12.4 - Prob. 12.39QAPCh. 12.4 - Prob. 12.40QAPCh. 12.4 - Prob. 12.41QAPCh. 12.4 - Prob. 12.42QAPCh. 12.4 - Prob. 12.43QAPCh. 12.4 - Prob. 12.44QAPCh. 12.4 - Prob. 12.45QAPCh. 12.4 - Prob. 12.46QAPCh. 12.4 - Prob. 12.47QAPCh. 12.4 - Prob. 12.48QAPCh. 12.4 - Prob. 12.49QAPCh. 12.4 - Prob. 12.50QAPCh. 12.5 - To make tomato soup, you add one can of water to...Ch. 12.5 - Prob. 12.52QAPCh. 12.5 - Prob. 12.53QAPCh. 12.5 - Prob. 12.54QAPCh. 12.5 - Prob. 12.55QAPCh. 12.5 - Prob. 12.56QAPCh. 12.5 - Prob. 12.57QAPCh. 12.5 - Prob. 12.58QAPCh. 12.5 - Prob. 12.59QAPCh. 12.5 - Prob. 12.60QAPCh. 12.6 - Prob. 12.61QAPCh. 12.6 - Prob. 12.62QAPCh. 12.6 - Prob. 12.63QAPCh. 12.6 - Prob. 12.64QAPCh. 12.6 - Prob. 12.65QAPCh. 12.6 - Prob. 12.66QAPCh. 12.7 - Prob. 12.67QAPCh. 12.7 - Prob. 12.68QAPCh. 12.7 - In each pair, identify the solution that will have...Ch. 12.7 - Prob. 12.70QAPCh. 12.7 - Prob. 12.71QAPCh. 12.7 - Prob. 12.72QAPCh. 12.7 - Prob. 12.73QAPCh. 12.7 - Prob. 12.74QAPCh. 12.8 - A 10% (m/v)starch solution is separated from a 1%...Ch. 12.8 - Prob. 12.76QAPCh. 12.8 - Prob. 12.77QAPCh. 12.8 - Prob. 12.78QAPCh. 12.8 - Prob. 12.79QAPCh. 12.8 - Prob. 12.80QAPCh. 12.8 - Each of the following mixtures is placed in a...Ch. 12.8 - Prob. 12.82QAPCh. 12 - Prob. 12.83FUCh. 12 - Prob. 12.84FUCh. 12 - Prob. 12.85FUCh. 12 - Prob. 12.86FUCh. 12 - Prob. 12.87UTCCh. 12 - Prob. 12.88UTCCh. 12 - Prob. 12.89UTCCh. 12 - Prob. 12.90UTCCh. 12 - Prob. 12.91UTCCh. 12 - Prob. 12.92UTCCh. 12 - Prob. 12.93UTCCh. 12 - Prob. 12.94UTCCh. 12 - Prob. 12.95UTCCh. 12 - Prob. 12.96UTCCh. 12 - Prob. 12.97AQAPCh. 12 - Prob. 12.98AQAPCh. 12 - Prob. 12.99AQAPCh. 12 - Prob. 12.100AQAPCh. 12 - Prob. 12.101AQAPCh. 12 - Prob. 12.102AQAPCh. 12 - Prob. 12.103AQAPCh. 12 - Prob. 12.104AQAPCh. 12 - If NaCl has a solubility of 36.0 g in 100. g of...Ch. 12 - Prob. 12.106AQAPCh. 12 - Prob. 12.107AQAPCh. 12 - Prob. 12.108AQAPCh. 12 - Prob. 12.109AQAPCh. 12 - Prob. 12.110AQAPCh. 12 - Prob. 12.111AQAPCh. 12 - Prob. 12.112AQAPCh. 12 - Prob. 12.113AQAPCh. 12 - What is the molarity of a solution containing 15.6...Ch. 12 - Prob. 12.115AQAPCh. 12 - Prob. 12.116AQAPCh. 12 - Prob. 12.117AQAPCh. 12 - Prob. 12.118AQAPCh. 12 - Prob. 12.119AQAPCh. 12 - Prob. 12.120AQAPCh. 12 - Prob. 12.121AQAPCh. 12 - Prob. 12.122AQAPCh. 12 - Prob. 12.123AQAPCh. 12 - Prob. 12.124AQAPCh. 12 - Prob. 12.125AQAPCh. 12 - Prob. 12.126AQAPCh. 12 - Prob. 12.127AQAPCh. 12 - Cadmium reacts with HCl to produce hydrogen gas...Ch. 12 - Prob. 12.129AQAPCh. 12 - Calculate the boiling point of each of the...Ch. 12 - Prob. 12.131AQAPCh. 12 - Prob. 12.132AQAPCh. 12 - Prob. 12.133CQCh. 12 - Write the net ionic equation to show the formation...Ch. 12 - Prob. 12.135CQCh. 12 - Prob. 12.136CQCh. 12 - Prob. 12.137CQCh. 12 - Prob. 12.138CQCh. 12 - Prob. 12.139CQCh. 12 - 9.134 A solution is prepared by dissolving 22.0 g...Ch. 12 - Prob. 12.141CQCh. 12 - Prob. 12.142CQCh. 12 - Prob. 12.143CQCh. 12 - Prob. 12.144CQCh. 12 - Prob. 12.145CQCh. 12 - Prob. 12.146CQ
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 7. Given: • The solubility of X at 100°C in water is 18.0 g/100 mL water. • The solubility of X at 0°C in water is 3.6 g/100 mL water. How many mL of boiling water would be required to dissolve 25g of X? If that solution was then cooled down to 0°C, how many grams of X could then crystallize out? What would be the maximum yield recovery for X?arrow_forwardD cent Solvent cvg.cengagenow.com The boiling point of water H₂O is 100.0 °C at 1 atmosphere. A nonvolatile, nonelectrolyte that dissolves in water is antifreeze (ethylene glycol). 9 [Review Topics] [References] Use the References to access important values if needed for this question. How many grams of antifreeze, CH₂OHCH₂OH (62.10 g/mol), must be dissolved in 291.0 grams of water to raise the boiling point by 0.500 °C? Refer to the table for the necessary boiling or freezing point constant. Kb (°C/m) Kf(°C/m) Water H₂O 0.512 Ethanol CH3CH₂OH 1.22 Chloroform CHC13 3.67 Benzene C6H6 2.53 Diethyl ether CH3 CH2 OCH2 CH3 2.02 Mass= Formula OWLV2 | Online teaching and learning resource from Cengage Learning 1.86 1.99 5.12 charrow_forward(14.6) Which of the following aqueous solution has the highest boiling point? O 1.00 M NaCl O 1.00 M Ca(NO3)2 O 1.00 M C6H1206 O 1.00 M NaNO3arrow_forward
- 8. (2 I. Consider the solubility graph below. II. Solubility (g solute/100 g H₂O) III. 1604 -140- 120 100+ 80 60+ 40- 20- 20 40 Temperature (C) 60 a. What is the concentration in molality of NaNO3 in a saturated solution at 60 °C? KNO₂ b. A solution contains 110 g of KNO3 in 100 g of H₂O at 60 °C and the temperature of the solution is cooled to 20 °C. How would the original solution be classified? [saturated, unsaturated, or supersaturated] NaNO, What mass of KNO3 will precipitate out of solution? Na SO NaCl How would the cooler solution be classified if precipitation occurs? [saturated, unsaturated, or supersaturated]arrow_forwardChemistry DJ 4.0 (11 11) How many moles of NaNO, will be left in solution at 20 °C if a solution at 60 °C in which there is 560 g of solute dissolved is left to cool? Volume of solution is 550 mL. (Solubility at 20 °C is 88 g per 100 g of H₂O). A) 6.59 B) 1.04 C) 0.09 D) 0.89arrow_forwardA 595 mL sample of table salt (NaCl) contains 28.6 g of NaCl. What is the molar concentration of the solution? (3 sf)arrow_forward
- (Q59) What is the molality of a solution that contains 73.7 grams of strontium fluoride that has been added to and dissolves in 3.13 liters of water? The density of water is 1.00 kg/L. (3 sf)arrow_forward10:24 1 Done Introductory+Chemistry ill a 10. What mass of Li₂O is needed to react with 1,060 g of CO₂? Li₂O(aq) + CO₂(g) → Li₂CO3(aq),arrow_forward7:22 Reset Done Send 2. A student dehydrated a sample of hydrate incompletely. Explain how the incomplete dehydration of the hydrate affects the calculation of the percent of water of hydration. What should the student do?arrow_forward
- Compound Lattice Energy (kJ/mol) NaF 930 MgF, 2978 The energy required to dissociate an ionic solid into gaseous ions (lattice energy) for the compounds NaF and MgF, is shown in the table above. On the basis of Coulomb's law, which of the following best helps to explain the large difference between the lattice energies of NaF and MgF, ? A The solubility of MgF, is less than that of NaF. B) The electronegativity of Mg is greater than that of Na. The mass of the Mg cation is greater than that of the Na cation. D The charge of the Mg cation is larger than that of the Na cation.arrow_forward5. At 25 "C, you make a homogeneous solution of 90.9 g of dextrose in 100 mL of water. Is this solution saturated, unsaturated, or supersaturated (13.2)? Saturated 6. If you then cool this solution to 5 °C, and the solution remains homogeneous, would the solution then be saturated, unsaturated, or supersaturated (13.2)? Supersaturated 7. Is DSW a saturated, unsaturated, or supersaturated solution of dextrose in water (13.2)? 8. Calculate the mole fraction of water in the solution described in Q5. The density of pure water at 25 °C is 0.997 g mL (13.3). 9. Calculate the molality of a solution of DSW (13.3). 10. Calculate the molarity of a solution of D5W (13.3). 11. Calculate the osmotic pressure Il of the solution of D5W (13.4). 2. Calculate the osmotic pressure II of the solution of dextrose from Q5 (13.4). 3. What would happen to your patient's blood if you administered an IV containing a solution of dextrose in water from Q5 (13.4)? Be specific. The osmotic pressure of human blood…arrow_forward#8 help pleasearrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning  Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:OpenStax

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning
Solutions: Crash Course Chemistry #27; Author: Crash Course;https://www.youtube.com/watch?v=9h2f1Bjr0p4;License: Standard YouTube License, CC-BY